
Exhibit 99.01

NASDAQ: TNXP © 2014 Tonix Pharmaceuticals Holding Corp. October 2014

Safe harbor s tatement Certain statements in this presentation regarding strategic plans, expectations and objectives for future operations or results are “forward - looking statements” as defined by the Private Securities Litigation Reform Act of 1995 . These statements may be identified by the use of forward - looking words such as "anticipate," "believe," "forecast," "estimate" and "intend," among others . These forward - looking statements are based on Tonix’s current expectations and actual results could differ materially . There are a number of factors that could cause actual events to differ materially from those indicated by such forward - looking statements . These factors include, but are not limited to, substantial competition ; our ability to continue as a going concern ; our need for additional financing ; uncertainties of patent protection and litigation ; uncertainties of government or third party payer reimbursement ; limited sales and marketing efforts and dependence upon third parties ; and risks related to failure to obtain U . S . Food and Drug Administration clearances or approvals and noncompliance with its regulations . As with any pharmaceutical under development, there are significant risks in the development, regulatory approval and commercialization of new products . The forward - looking statements in this presentation are made as of the date of this presentation, even if subsequently made available by the Company on its website or otherwise . Tonix does not undertake an obligation to update or revise any forward - looking statement, except as required by law . Investors should read the risk factors set forth in the amended Annual Report on Form 10 - K for the year ended December 31 , 2013 , as filed with the Securities and Exchange Commission (the “SEC”) on March 28 , 2014 and future periodic reports filed with the SEC on or after the date hereof . All of the Company's forward - looking statements are expressly qualified by all such risk factors and other cautionary statements . 2

Overview First - in - class medicines for common disorders of the central nervous system (CNS) Innovative treatment paradigms Late stage candidates Large unmet medical needs Unencumbered intellectual property portfolio Strong balance sheet, experienced team Multiple successes in drug approvals and value creation Well - capitalized to execute on key near - term milestones 3 Fibromyalgia Post - traumatic Stress Disorder Episodic Tension - type Headache
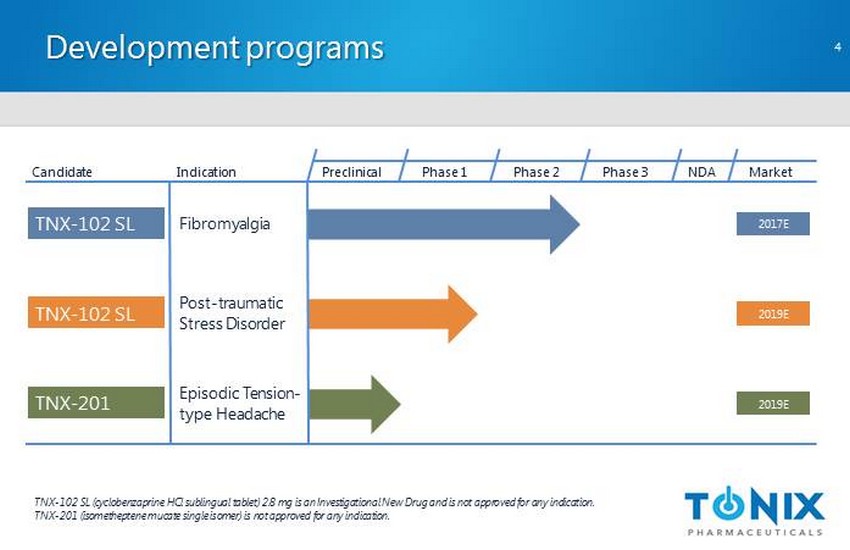
Development programs Preclinical Phase 1 NDA Market Candidate Indication TNX - 102 SL (cyclobenzaprine HCl sublingual tablet) 2.8 mg is an Investigational New Drug and is not approved for any indication. TNX - 201 (isometheptene mucate single isomer) is not approved for any indication. Phase 2 Phase 3 Post - traumatic Stress Disorder TNX - 102 SL 2019E TNX - 102 SL Fibromyalgia 2017E TNX - 201 Episodic Tension - type Headache 2019E 4

Targeting sleep quality in fibromyalgia (FM) and post - traumatic stress disorder (PTSD) Non - restorative sleep linked to pain, fatigue, hyper - vigilance, and arousals Restorative sleep improves symptoms of fibromyalgia and PTSD TNX - 102 SL is designed as a chronic therapy for bedtime use Novel molecular target for tension - type headache Based on proprietary discoveries by Tonix Mechanism of action distinct from acetaminophen or barbiturates New approaches to treating CNS disorders 5
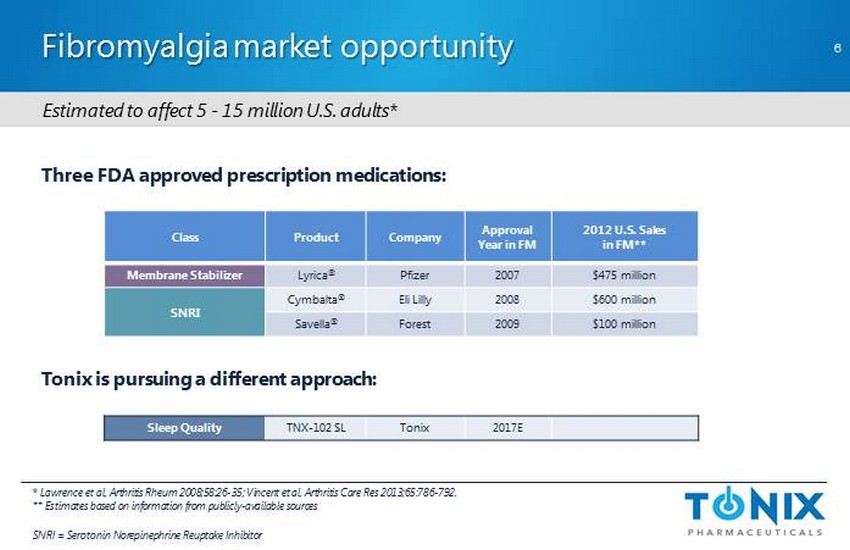
CONFIDENTIAL Fibromyalgia market opportunity * Lawrence et al, Arthritis Rheum 2008;58:26 - 35; Vincent et al, Arthritis Care Res 2013;65:786 - 792. ** Estimates based on information from publicly - available sources SNRI = Serotonin Norepinephrine Reuptake Inhibitor Three FDA approved prescription medications: Tonix is pursuing a different approach: Class Product Company Approval Year in FM 2012 U.S. S ales in FM** Membrane Stabilizer Lyrica ® Pfizer 2007 $475 million SNRI Cymbalta ® Eli Lilly 2008 $600 million Savella ® Forest 2009 $100 million Sleep Quality TNX - 102 SL Tonix 2017E 6 E stimated to affect 5 - 15 million U.S. adults*

Fibromyalgia treatments dissatisfy many patients Chronic , widespread pain with sleep, fatigue, mood, and memory problems Poor quality of life Typical patient has onset at 30 - 40 years of age with persistence for rest of life Patients remain unsatisfied despite approved products Patients typically take multiple medications ‘ Off - label’ use of opioids and sedative - hypnotics despite no sustained benefit Expensive, burdensome condition Health utilization and medication costs are substantial Managed care / payers recognize need for new medicines 7
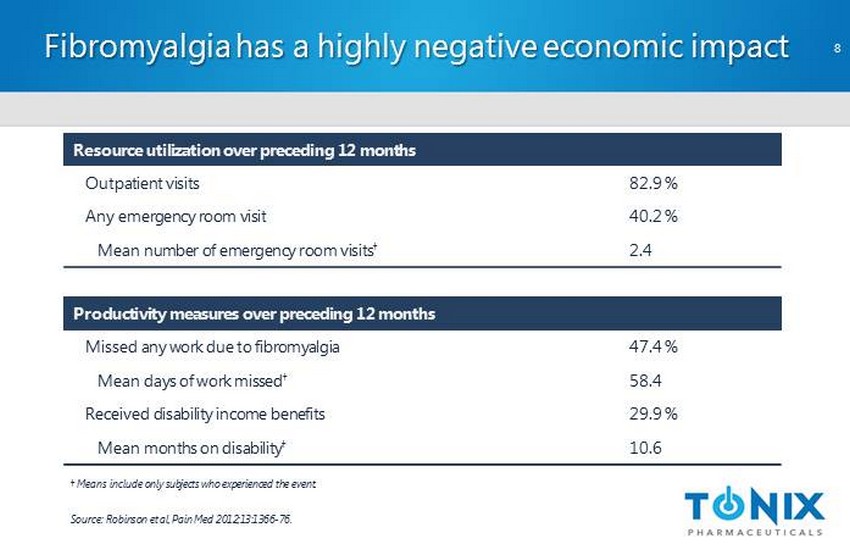
CONFIDENTIAL Fibromyalgia has a highly negative economic impact Source: Robinson et al, Pain Med 2012;13:1366 - 76. Resource utilization over preceding 12 months Outpatient visits 82.9 % Any emergency room visit 40.2 % Mean number of emergency room visits † 2.4 Productivity measures over preceding 12 months Missed any work due to fibromyalgia 47.4 % Mean days of work missed † 58.4 Received disability income benefits 29.9 % Mean months on disability † 10.6 † Means include only subjects who experienced the event. 8

CONFIDENTIAL Restorative sleep improves pain and other fibromyalgia symptoms >90% of fibromyalgia patients complain of poor sleep quality* Clinical validation from proof - of - concept study Phase 2a trial showed pain and sleep benefit with very low dose oral cyclobenzaprine (CBP)** TNX - 102 SL: sublingual tablet formulation of CBP Designed for chronic use at bedtime Phase 2b results demonstrate broad - spectrum activity in patients enrolled in the BESTFIT trial*** Significant improvements across core fibromyalgia symptom domains Well - tolerated * Swick , Ther Adv Musculoskel Dis 2011;3:167 - 178. ** Moldofsky et al., J Rheum 2011;38:2653 - 63. *** Phase 2b BESTFIT preliminary top line results Sleep quality is a new target for fibromyalgia therapy 9
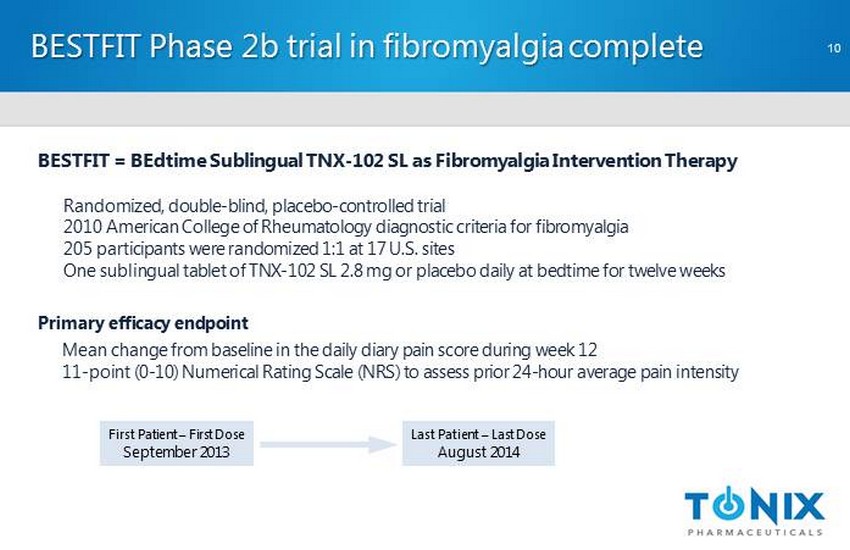
CONFIDENTIAL BESTFIT Phase 2b trial in fibromyalgia complete 10 BESTFIT = BEdtime Sublingual TNX - 102 SL as Fibromyalgia Intervention Therapy Randomized, double - blind, placebo - controlled trial 2010 American College of Rheumatology diagnostic criteria for fibromyalgia 205 participants were randomized 1:1 at 17 U.S. sites One sublingual tablet of TNX - 102 SL 2.8 mg or placebo daily at bedtime for twelve weeks Primary efficacy endpoint M ean change from baseline in the daily diary pain score during week 12 11 - point (0 - 10) Numerical Rating Scale (NRS) to assess prior 24 - hour average pain intensity First Patient – First Dose September 2013 Last Patient – Last Dose August 2014

CONFIDENTIAL TNX - 102 SL improved pain in fibromyalgia in the BESTFIT study 11 Outcome Measure at Week 12 Intent - to - Treat Population † p value Method Daily Pain Diary, NRS Mean Change** 0.086 0.172 MMRM JTC - MI Daily Pain Diary, NRS Proportion Achieving 30% Improvement* 0.030 LR Clinic NRS 7 - day pain recall Mean Change 0.029 MMRM FIQ - R Pain Item Mean Change 0.004 MMRM NRS = Numeric Pain Scale; FIQ - R = Fibromyalgia Impact Questionnaire - Revised MMRM = Mixed - Effect Model Repeated Measure; JTC - MI = Jump to Control - Multiple Imputation (FDA - preferred analysis); LR = Logistic Regression ** Declared primary endpoint; was primary endpoint for FDA approvals of Lyrica and Cymbalta * Declared secondary endpoint; was primary endpoint for FDA approval of Savella † N=205 (TNX - 102 SL N=103, placebo N=102) p < 0.05 ; a chieved statistical significance Source: Phase 2b BESTFIT preliminary top line results

CONFIDENTIAL TNX - 102 SL improved sleep quality in fibromyalgia in the BESTFIT study 12 Outcome Measure at Week 12 Intent - to - Treat Population p value Method Daily Sleep Quality Diary, NRS Mean Change* <0.001 MMRM PROMIS Sleep Disturbance T - score Change* 0.003 0.004 MMRM JTC - MI FIQ - R Sleep Quality Item Mean Change <0.001 MMRM PROMIS = Patient - Reported Outcome Measures in Sleep * Declared secondary endpoint Source: Phase 2b BESTFIT preliminary top line results p < 0.05 ; a chieved statistical significance
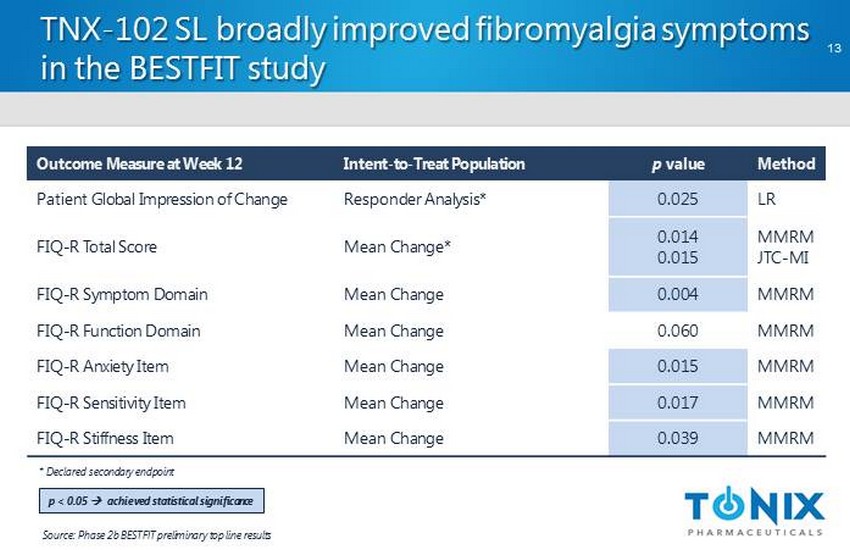
CONFIDENTIAL TNX - 102 SL broadly improved fibromyalgia symptoms in the BESTFIT study 13 Outcome Measure at Week 12 Intent - to - Treat Population p value Method Patient Global Impression of Change Responder Analysis* 0.025 LR FIQ - R Total Score Mean Change* 0.014 0.015 MMRM JTC - MI FIQ - R Symptom Domain Mean Change 0.004 MMRM FIQ - R Function Domain Mean Change 0.060 MMRM FIQ - R Anxiety Item Mean Change 0.015 MMRM FIQ - R Sensitivity Item Mean Change 0.017 MMRM FIQ - R Stiffness Item Mean Change 0.039 MMRM Source: Phase 2b BESTFIT preliminary top line results * Declared secondary endpoint p < 0.05 ; a chieved statistical significance
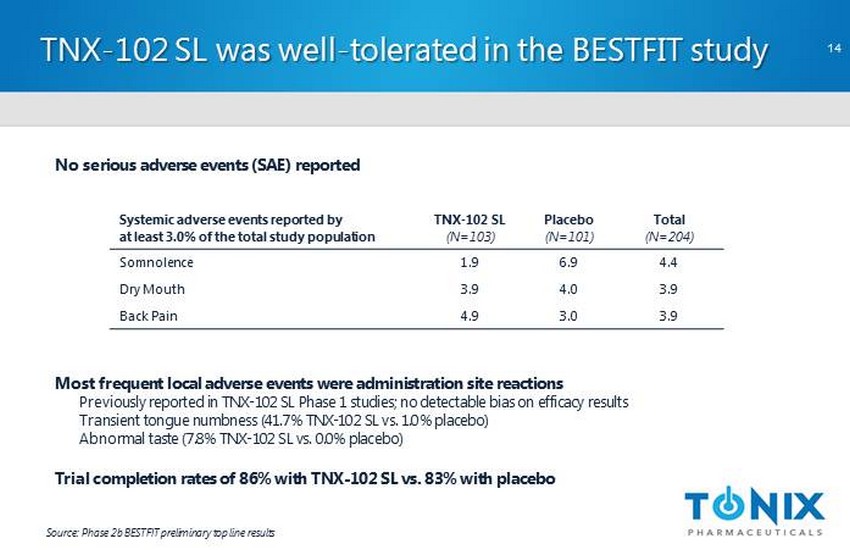
CONFIDENTIAL TNX - 102 SL was well - tolerated in the BESTFIT study 14 Source: Phase 2b BESTFIT preliminary top line results No serious adverse events (SAE) reported Most frequent local adverse events were administration site reactions Previously reported in TNX - 102 SL Phase 1 studies; no detectable bias on efficacy results Transient tongue numbness (41.7% TNX - 102 SL vs. 1.0% placebo) Abnormal taste (7.8% TNX - 102 SL vs. 0.0% placebo) Trial completion rates of 86% with TNX - 102 SL vs. 83% with placebo Systemic adverse events reported by at least 3.0% of the total study population TNX - 102 SL (N=103) Placebo (N=101) Total (N=204) Somnolence 1.9 6.9 4.4 Dry Mouth 3.9 4.0 3.9 Back Pain 4.9 3.0 3.9

CONFIDENTIAL Acceptable to submit 505(b)( 2) New Drug Application (NDA) Two adequate and well - controlled efficacy and safety trials in fibromyalgia patients Requesting FDA meeting to review BESTFIT results and discuss efficacy requirements for NDA One - year exposure data to support chronic use label Open - label extension study to BESTFIT ongoing, to complete in August 2015 Definitive repeat dose pharmacokinetic “bridging ” study Registration program for TNX - 102 SL in fibromyalgia 15 End - of - Phase 2 clinical/non - clinical meeting held with FDA in February 2013
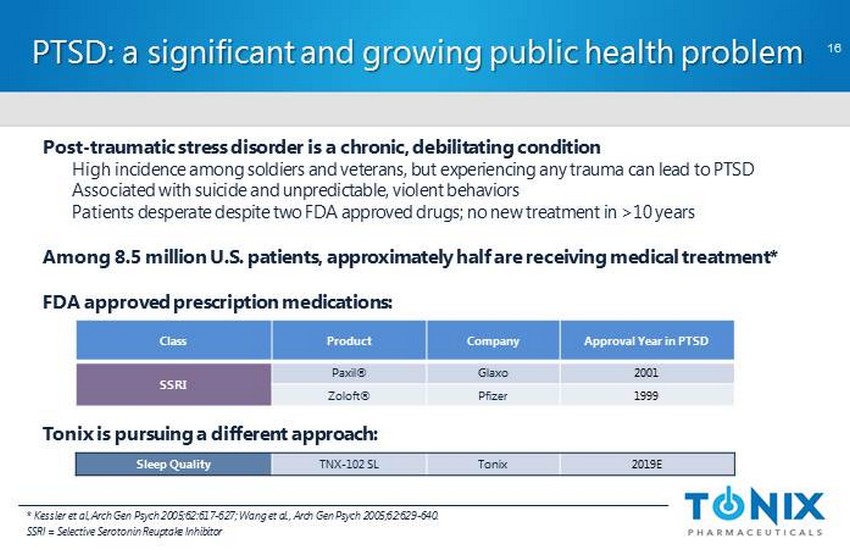
CONFIDENTIAL Post - traumatic stress disorder is a chronic, debilitating condition High incidence among soldiers and veterans, but experiencing any trauma can lead to PTSD Associated with suicide and unpredictable, violent behaviors Patients desperate despite two FDA approved drugs; no new treatment in >10 years Among 8.5 million U.S. patients, approximately half are receiving medical treatment * FDA approved prescription medications: Tonix is pursuing a different approach: PTSD: a significant and growing public health problem 16 Class Product Company Approval Year in PTSD SSRI Paxil ® Glaxo 2001 Zoloft ® Pfizer 1999 Sleep Quality TNX - 102 SL Tonix 2019E * Kessler et al, Arch Gen Psych 2005;62:617 - 627; Wang et al., Arch Gen Psych 2005;62:629 - 640. SSRI = Selective Serotonin Reuptake Inhibitor

CONFIDENTIAL PTSD patients complain of poor sleep quality as a core symptom Distressing dreams (nightmares) are part of “re - experiencing” Restless sleep is part of “hyper - arousal” Poor sleep quality after trauma is linked to onset of PTSD Correlated with depression, substance abuse and suicide TNX - 102 SL targets mechanisms associated with treating disturbed sleep in PTSD Antagonist at serotonin 2A, adrenergic α - 1, histamine - 1 receptors Sleep quality is a new target for PTSD therapy 17

CONFIDENTIAL PTSD program with TNX - 102 SL 18 P hase 2 efficacy study of TNX - 102 SL to begin in 4Q 2014 Leverage fibromyalgia formulation, clinical experience, manufacturing know - how Overlap between PTSD and fibromyalgia ~50% of fibromyalgia or PTSD patients meet criteria for the other disorder Patients experience disturbed sleep Widespread pain is considered “co - morbid” with PTSD Opioid and sedative - hypnotic drug misuse common

CONFIDENTIAL “AtEase” Phase 2 trial of TNX - 102 SL in PTSD Randomized , double - blind, placebo - controlled trial N=220; approximately 25 U.S. clinical sites Primary efficacy endpoint: D ifference in Clinician - Administered PTSD Scale (CAPS) score between TNX - 102 SL 2.8 mg and placebo TNX - 102 SL at bedtime once - daily Placebo at bedtime once - daily 12 weeks N = 88 19 To begin enrollment in 4Q14 TNX - 102 SL at bedtime once - daily N = 88 N = 44 2.8 mg 5.6 mg open - label extension
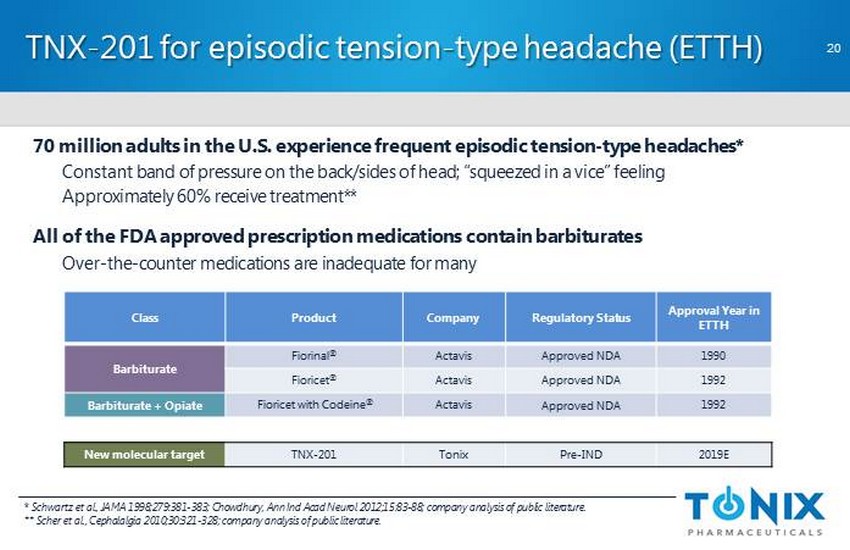
CONFIDENTIAL TNX - 201 for episodic tension - type headache (ETTH) * Schwartz et al., JAMA 1998;279:381 - 383; Chowdhury, Ann Ind Acad Neurol 2012;15:83 - 88; company analysis of public literature. ** Scher et al., Cephalalgia 2010;30:321 - 328; company analysis of public literature. 70 million adults in the U.S. experience frequent episodic tension - type headaches* Constant band of pressure on the back/sides of head; “squeezed in a vice” feeling Approximately 60% receive treatment** All of the FDA approved prescription medications contain barbiturates Over - the - counter medications are inadequate for many Class Product Company Regulatory Status Approval Year in ETTH Barbiturate Fiorinal ® Actavis Approved NDA 1990 Fioricet ® Actavis Approved NDA 1992 Barbiturate + Opiate Fioricet with Codeine ® Actavis Approved NDA 1992 New molecular target TNX - 201 Tonix Pre - IND 2019E 20

CONFIDENTIAL TNX - 201 to enter clinical development in 2015 Novel molecular mechanism Based on proprietary discoveries by Tonix Non - barbiturate, non - opioid Mechanism of action distinct from acetaminophen and barbiturates Comparative pharmacokinetic and safety study to be conducted in 1Q 2015 Pre - IND meeting with FDA held in January 2014 21

CONFIDENTIAL Intellectual property Composition - of - matter (eutectic) Patents filed Protection expected to 2034 Pharmacokinetics (PK) Patents filed Protection expected to 2033 Method - of - use Fibromyalgia: patents issued, 3Q 2020 expiry PTSD: patents filed TNX - 102 SL Fibromyalgia, PTSD TNX - 201 H eadache Composition - of - matter (isomer) Patents filed Protection expected to 2033 All IP wholly - owned by Tonix without obligations to others 22

CONFIDENTIAL Milestones – recent and upcoming Corporate □ July 2014 – $7.2 million in net proceeds from common stock offering TNX - 102 SL – Fibromyalgia □ September 2014 – Reported top line results from Phase 2b BESTFIT study □ Meet with FDA to determine remaining requirements for NDA □ Begin Phase 3 registration program TNX - 102 SL – Post - traumatic Stress Disorder □ June 2014 – Received IND clearance □ 4Q 2014 – Start Phase 2 AtEase efficacy study TNX - 201 – Episodic Tension - type Headache □ January 2014 – Held Pre - IND meeting □ 1Q 2015 – Conduct clinical pharmacology study x x x x 23

CONFIDENTIAL Bruce Daugherty, PhD Chief Scientific Officer Seth Lederman, MD Chief Executive Officer Management team Leland Gershell, MD, PhD Chief Financial Officer Don Kellerman, PharmD SVP, Clinical Development & Regulatory Affairs 24

CONFIDENTIAL John Rhodes NYSERDA, NRDC Booz Allen Hamilton Donald Landry, MD, PhD Chair, Department of Medicine Columbia University Stuart Davidson Alkermes Combion Seth Lederman, MD (Chair) Targent Pharmaceuticals Vela Pharmaceuticals Samuel Saks, MD ALZA Jazz Pharmaceuticals Patrick Grace WR Grace Chemed Ernest Mario, PhD Glaxo, ALZA Reliant Pharmaceuticals Board of directors Charles Mather Janney Montgomery Scott Cowen, Smith Barney 25

CONFIDENTIAL Financial summary NASDAQ: TNXP Cash reported at June 30 , 2014* $ 43.9 million Net cash used in operations in 2Q14 $ 5.7 million Shares outstanding † 10.8 million * Does not include $7.2 million in net proceeds from stock offering in July 2014. † As of October 8, 2014 26

CONFIDENTIAL Opportunity summary TNX - 102 SL: late - stage clinical program for large market opportunity Strong clinical evidence of broad activity in lead indication Well - known safety profile with excellent tolerability in patients studied Current fibromyalgia treatment options leave many patients unsatisfied Multiple opportunities (fibromyalgia , PTSD, headache) Team distinguished by track record of successful drug development Well - capitalized to execute on key near - term milestones 27

© 2014 Tonix Pharmaceuticals Holding Corp. NASDAQ: TNXP