
Tonix Pharmaceuticals Holding Corp. 8-K
Exhibit 99.02

© 2023 Tonix Pharmaceuticals Holding Corp. TNX - 601 ER Major Depressive Disorder NASDAQ: TNXP Version P04 39 May 1, 2023 (Doc 11 98 )

2 © 2023 Tonix Pharmaceuticals Holding Corp. Cautionary Note on Forward - Looking Statements Certain statements in this presentation regarding strategic plans, expectations and objectives for future operations or results are “forward - looking statements” as defined by the Private Securities Litigation Reform Act of 1995. These statements may be identified by the use of forward - looking words such as “anticipate,” “believe,” “forecast,” “estimate” and “intend,” among others. These forward - looking statements are based on Tonix’s current expectations and actual results could differ materially. There are a number of factors that could cause actual events to differ materially from those indicated by such forward - looking statements. These factors include, but are not limited to, the risks related to failure to obtain FDA clearances or approvals and noncompliance with FDA regulations; delays and uncertainties caused by the global COVID - 19 pandemic; risks related to the timing and progress of clinical development of our product candidates; our need for additional financing; uncertainties of patent protection and litigation; uncertainties of government or third party payor reimbursement; limited research and development efforts and dependence upon third parties; and substantial competition. As with any pharmaceutical under development, there are significant risks in the development, regulatory approval and commercialization of new products. The forward - looking statements in this presentation are made as of the date of this presentation, even if subsequently made available by Tonix on its website or otherwise. Tonix does not undertake an obligation to update or revise any forward - looking statement, except as required by law. Investors should read the risk factors set forth in the Annual Report on Form 10 - K for the year ended December 31, 2022, as filed with the Securities and Exchange Commission (the “SEC”) on March 13, 2023, and periodic reports and current reports filed with the SEC on or after the date thereof. All of Tonix's forward - looking statements are expressly qualified by all such risk factors and other cautionary statements.

3 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO PROFILE DEVELOPMENT PROGRAM Patents Issued TNX - 601 E R*: Depression Tianeptine Hemioxalate Extended - Release Tablets (39.4 mg) CNS PORTFOLIO • A novel, oral, extended - release once - daily tablet • Treatment effect of tianeptine sodium immediate release t.i.d. in depression is well - established • Tianeptine restores neuroplasticity in animal models • Indirectly modulates the glutamatergic system • Does not interact with AMPA, NMDA or kainate receptors 1 Differentiators: Relative to tianeptine IR available ex - US: • Once daily dosing Relative to traditional antidepressants: • Unique mechanism of action – beyond neurotransmitter modulation • Tianeptine sodium IR has similar efficacy but fewer side effects than traditional antidepressants Market Entry: Major Depressive Disorder (MDD) Additional Indications: PTSD, Neurocognitive Disorder From Corticosteroids, Alzheimer’s Disease 2 Status: Phase 2 MDD study UPLIFT is currently enrolling Next Steps: Interim analysis results on first 50% of sample expected 4Q 2023 1 AMPA= α - amino - 3 - hydroxy - 5 - methyl - 4 - isoxazolepropionic acid; NMDA=N - methyl - D - aspartate 2 García - Alberca et al., 2022. J Alzheimers Dis . 88(2):707 - 720 *TNX - 601 ER has not been approved for any indication.
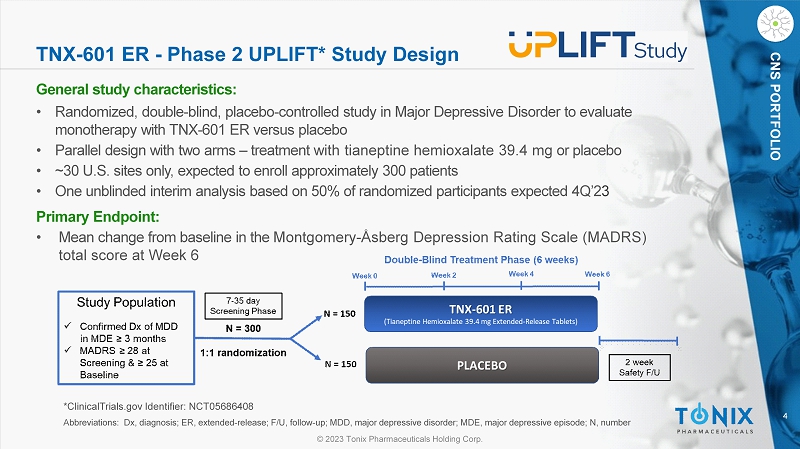
© 2023 Tonix Pharmaceuticals Holding Corp. 4 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO TNX - 601 ER - Phase 2 UPLIFT* Study Design General s tudy c haracteristics: • Randomized, double - blind, placebo - controlle d study in Major Depressive Disorder to evaluate monotherapy with TNX - 601 ER versus placebo • Parallel design with two arms – treatment with tianeptine hemioxalate 39.4 mg or placebo • ~30 U.S. sites only, expected to enroll approximately 300 patients • One unblinded interim analysis based on 50% of randomized participants expected 4Q’2 3 Primary Endpoint: • Mean change from baseline in the Montgomery - Åsberg Depression Rating Scale (MADRS) total score at W eek 6 *ClinicalTrials.gov Identifier: NCT05686408 Abbreviations: Dx, diagnosis; ER, extended - release; F/U, follow - up; MDD, major depressive disorder; MDE, major depressive episo de; N, number

© 2023 Tonix Pharmaceuticals Holding Corp. 5 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Major Depressive Disorder (MDD) • Major depressive disorder (MDD) is a leading cause of disability worldwide, with 21 million adults in the US alone experiencing a depressive episode in 2020 1 • L ifetime prevalence of 16%, and associated with important psychological suffering, as well as elevated rates of suicide and worse prognosis of comorbid medical conditions 2,3 • Highly comorbid with other psychiatric disorders, e.g., anxiety disorders, substance use disorders, as well as medical conditions, e.g., cardiovascular disease, metabolic syndromes, respiratory diseases, various deficiencies, infections, collagen disorders, endocrine diseases, etc. • Occurs in women at three times the rate in men • Hormonal aspects can significantly impact course and treatment (especially evident in post - partum depression) • Increased incidence during COVID - 19 pandemic in all age groups and both sexes • Most treatment guidelines support use of antidepressants in moderate to severe MDD Epidemiology and Characteristics of Depression 1 Substance Abuse and Mental Health Services Administration (SAMHSA). 2020. Key Substance Use and Mental Health Indicators in t he United States: Results from the 2020 National Survey on Drug Use and Health. 2 Kupfer et al., 2012. The Lancet . 379, 1045 – 1055 3 Otte et al., 2016. Nat. Rev. Dis. Primer . 2:16065

© 2023 Tonix Pharmaceuticals Holding Corp. 6 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO • The Sequenced Treatment Alternatives to Relieve Depression (STAR*D) study, regarded as the largest antidepressant trial ever conducted, indicated approximately 30% of depressed patients fail to achieve remission, even after multiple treatment attempts 1,2 • SSRIs are currently the most prescribed class of antidepressants, yet only about 50% of patients with MDD respond to initial SSRI treatment , and only 35 - 40% of those patients achieve full remission 1 • Antidepressant treatments often continue for years, and the side effect profiles of the monoaminergic antidepressants are intolerable to many • There is a high unmet need for new classes of antidepressants with different mechanisms of action High Unmet N eed for New C lasses of Antidepressants 1 Rush et al., 2006. Am J Psychiatry. 163:1905 – 1917 2 Rush et al., 2004. Control Clin Trials . 25(1):119 - 42

© 2023 Tonix Pharmaceuticals Holding Corp. 7 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO About TNX - 601 ER CNS PORTFOLIO • Tianeptine sodium (amorphous) immediate release (IR) tablets have been available in Europe and many countries in Asia and Latin America for the treatment of MDD since it was first marketed in France in 1989 • Due to its short half - life, tianeptine sodium IR is taken three times daily, which is challenging for patient adherence • Currently, there is no tianeptine - containing product approved in the U.S. and no extended - release tianeptine product approved anywhere in the world • Tonix discovered a novel hemioxalate salt of tianeptine that may provide improved stability, consistency, and manufacturability compared to known salt forms of tianeptine • TNX - 601 ER is taken once daily, increasing patient adherence and is thereby anticipated to improve the overall effectiveness of treatment compared to that of tianeptine sodium IR Targeted therapy for Major Depressive Disorder

© 2023 Tonix Pharmaceuticals Holding Corp. 8 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Clinical Trials of Tianeptine Sodium • Antidepressant efficacy confirmed in multicenter double - blind, placebo - controlled, randomized trials 1,2 • Enriched enrollment randomized withdrawal design trial of long - term (16.5 months) treatment demonstrated reduction of MDD relapse and recurrence by 2 - to 3 - fold compared to placebo 3 • Head - to - head comparisons showing equivalent efficacy of tianeptine with: ‒ TCAs ▪ Imipramine 1 ▪ Amitriptyline 4,5,6 ‒ SSRIs ▪ Fluoxetine 4,7 ▪ Sertraline 8 ▪ Paroxetine 9,10,11 ▪ Escitalopram 12 ‒ Mianserin 13 • Rigorous meta - analysis 14,15 of studies comparing tianeptine to SSRIs concluded tianeptine at least as effective as SSRIs, and trend noted for better overall acceptability profile in treatment of depressed patients Placebo - controlled and comparative trials in depression 1 Cassono et al., 1996. Eur Psychiatry. 11(5):254 - 9 2 Costa e Silva et al., 1997. Neuropsychobiology . 35(1):24 - 9 3 Dalery et al., 2001. Hum Psychopharmacol .16(S1):S39 - S47 4 Lôo et al., 1999. Neuropsychobiology .19(2):79 - 85 5 Guelfi et al., 1989. Neuropsychobiology . 22(1):41 - 8 11 Nickel et al., 2003. J Clin Psychopharmacol . 23(2):155 - 68 12 Emsley et al., 2018. J Clin Psychiatry. 79(4):17m11741 13 Brion et al., 1996. Presse Med . 25(9):461 - 8 14 Kasper et al., 2002. Eur Psychiatry . 17 Suppl 3:331 - 40 15 Olié et al., 2003. Encephale . 29(4 Pt 1):322 - 8 6 Invernizzi et al., 1994. Neuropsychobiology . 30(2 - 3):85 - 93 7 Novotny et al., 2002. Hum Psychopharmacol . 17(6):299 - 303 8 Szádóczky et al., 2002. Encephale . 28(4):343 - 9 9 Lepine et al., 2001. Hum Psychopharmacol . 16(3):219 - 227 10 Waintraub et al., 2002. CNS Drugs . 16(1):65 - 75
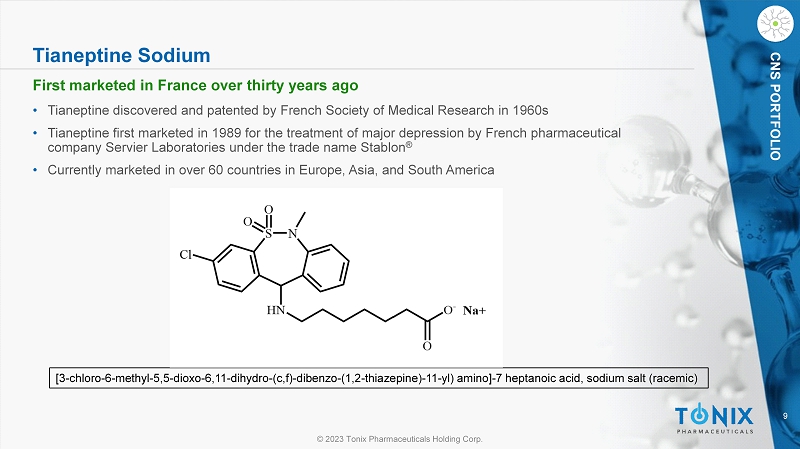
© 2023 Tonix Pharmaceuticals Holding Corp. 9 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Tianeptine Sodium • Tianeptine discovered and patented by French Society of Medical Research in 1960s • Tianeptine first marketed in 1989 for the treatment of major depression by French pharmaceutical company Servier Laboratories under the trade name Stablon ® • Currently marketed in over 60 countries in Europe, Asia, and South America [3 - chloro - 6 - methyl - 5,5 - dioxo - 6,11 - dihydro - ( c,f ) - dibenzo - (1,2 - thiazepine) - 11 - yl) amino] - 7 heptanoic acid, sodium salt (racemic) First marketed in France over thirty years ago
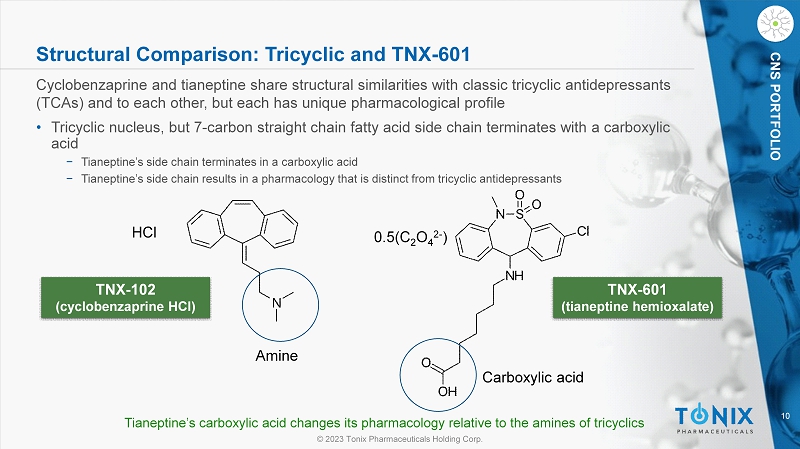
© 2023 Tonix Pharmaceuticals Holding Corp. 10 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Structural Comparison: Tricyclic and TNX - 601 Cyclobenzaprine and tianeptine share structural similarities with classic tricyclic antidepressants (TCAs) and to each other, but each has unique pharmacological profile • Tricyclic nucleus, but 7 - carbon straight chain fatty acid side chain terminates with a carboxylic acid − Tianeptine’s side chain terminates in a carboxylic acid − Tianeptine’s side chain results in a pharmacology that is distinct from tricyclic antidepressants TNX - 102 (cyclobenzaprine HCl) TNX - 601 (tianeptine hemioxalate ) HCl 0.5(C 2 O 4 2 - ) Tianeptine’s carboxylic acid changes its pharmacology relative to the amines of tricyclics Carboxylic acid Amine

© 2023 Tonix Pharmaceuticals Holding Corp. 11 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Proposed Mechanism of Action (MOA) • The proposed MOA of TNX - 601 ER is distinct from traditional monoaminergic antidepressants in the U.S. − “It is now recognized that monoamine deficits are only part of the story and are not sufficient on their own to explain the mechanism of action of antidepressants” 1 • MDD may be associated with an impairment of neuroplasticity and cellular resilience, and antidepressant medications may act by normalizing this impairment 2 - 4 • In animal studies involving severe stress exposure, TNX - 601 ER has a unique MOA that restores brain neuroplasticity by exerting biological effects on neurons and glial cells that increase arborization of dendrites in critical hippocampal circuitry 1 • In animal models, tianeptine also reverses stress - induced impairments in synaptic glutamate neurotransmission, and it restores hippocampal neurogenesis 1 1 McEwen et al., 2010. Mol. Psychiatry . 15(3), 237 – 249 . 2 Duman et al., 1999. Biol Psychiatry . 46: 1181 – 1191. 3 Manji et al., 2001. Psychopharmacol Bull. 35: 5 – 49. 4 Pittenger et al., 2008. N europsychopharmacology. 33: 88 – 109. . Effects on Neuroplasticity & Neurogenesis
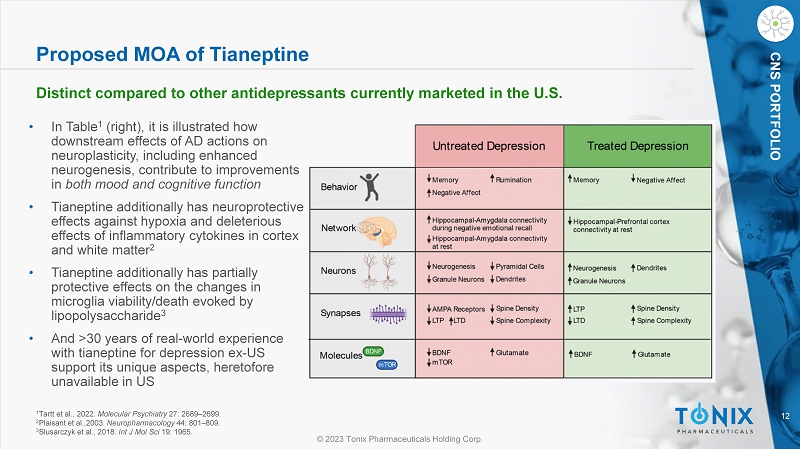
© 2023 Tonix Pharmaceuticals Holding Corp. 12 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO 1 Tartt et al., 2022. Molecular Psychiatry 27: 2689 – 2699. 2 Plaisant et al.,2003. Neuropharmacology 44: 801 – 809. 3 Slusarczyk et al., 2018. Int J Mol Sci 19: 1965. • In Table 1 (right), it is illustrated how downstream effects of AD actions on neuroplasticity, including enhanced neurogenesis, contribute to improvements in both mood and cognitive function • Tianeptine additionally has neuroprotective effects against hypoxia and deleterious effects of inflammatory cytokines in cortex and white matter 2 • Tianeptine additionally has partially protective effects on the changes in microglia viability/death evoked by lipopolysaccharide 3 • And >30 years of real - world experience with tianeptine for depression ex - US support its unique aspects, heretofore unavailable in US Proposed MOA of Tianeptine Distinct compared to other antidepressants currently marketed in the U.S.

© 2023 Tonix Pharmaceuticals Holding Corp. 13 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Observations that Relate Tianeptine’s Action to µ - Opioid Receptors • In 2014, tianeptine was reported to be a weak µ – opioid agonist by Javitch & Sames at Columbia 1 − K i = 383 nM and EC 50 = 194 nM 1 − Others have found even lower binding and activity, e.g., K i = 768 nM 2 or EC 50 >3 uM 3 • In 2017, tianeptine’s µ – opioid activity was implicated as central to its mechanism of treating depression by Hen, Javitch & Sames at Columbia 4,5 − Observations: e.g., The effect of tianeptine at 30 mg/kg on the Porsolt Forced Swim Test (FST) was decreased by naloxone treatment or in knock - out mice lacking the µ – opioid receptor − Tonix interpretation : While Samuels et al . provided information on the effects of high doses of tianeptine in murine analgesic models, the presented FST studies did not conclusively show the antidepressant effect of tianeptine at the therapeutic dose in humans requires µ - opioid receptor agonism 1 Gassaway et al., 2014. Transl Psychiatry . 4(7):e411 2 BL Roth PDSP K i database; https://pdsp.unc.edu/databases 3 Vandeputte et al., 2020. Arch Toxicol . 94(11):3819 - 3830 4 Samuels et al., 2017. Neuropsychopharmacology . 42(10):2052 - 2063 5 Han et al., 2022. Neuropsychopharmacology . 47(7):1387 - 1397 Tianeptine is a weak µ - opiate receptor agonist

© 2023 Tonix Pharmaceuticals Holding Corp. 14 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Tianeptine's off - target activity • Based on these µ – opioid data and interpretations, unregulated tianeptine entered the US ‒ As a research chemical - not for human use ‒ As an ingredient in food supplements sold over the counter ‒ Without any submitted data or regulatory status, promoted as a “smart drug” (nootropic) sold over the internet • Because of low affinity binding and agonist activity on µ - opioid receptor, there is the potential abuse liability of tianeptine drug substance when available in large quantities by ‒ People seeking a µ - opioid “high” ‒ People self - managing withdrawal effects from opioids Illicit or unregulated introduction of the drug substance to the United States

© 2023 Tonix Pharmaceuticals Holding Corp. 15 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Prescription Tianeptine has Low Incidence of Abuse in France • Tianeptine and its MC5 metabolite are weak mu - opioid (µ - opioid) receptor (MOR) agonists 1 that present a potential abuse liability if illicitly misused in large quantities (typically abused at 8 - 80 times the therapeutic dose on a daily basis 2 ). • In patients who were prescribed tianeptine for depression, the French Transparency Committee found a low incidence of misuse ‒ Approximately 1 case per 1,000 patients treated 3 suggesting low abuse liability when used at the antidepressant dose in patients prescribed tianeptine for depression. • Clinical trials have shown that abrupt cessation of a therapeutic course of tianeptine does not appear to result in dependence or withdrawal symptoms following treat ment for : ‒ 6 - weeks 4 - 8 ‒ 3 - months 9 ‒ 12 - months 10 Low activity at µ - opioid receptor is associated with low misuse of prescription oral tianeptine 1 Gassaway et al., 2014. Transl Psychiatry . 4(7):e411 2 Lauhan et al., 2018. Psychosomatics. 59(6), 547 – 53 3 Haute Authorite de Sante. Transparency Committee Opinion. Stablon 12.5 Mg, Coated Tablet, Re - Assessment of Actual Benefit at the Request of the Transparency Committee. December 5, 2012. 4 Emsley et al., 2018. J. Clin. Psychiatry . 79 (4) 5 Bonierbale et al., 2003. Curr Med Res Opin . 19(2):114 - 124 6 Guelfi et al., 1989. Neuropsychobiology . 22 (1), 41 – 48 7 Invernizzi et al., 1994. Neuropsychobiology . 30 (2 – 3), 85 – 93 8 Lepine et al., 2001. Hum. Psychopharmacol . 16 (3), 219 – 227 9 Guelfi et al., 1992. Neuropsychobiology . 25 (3), 140 – 148. 10 Lôo et al., 1992. Br. J. Psychiatry. Suppl. No. 15, 61 – 65.

© 2023 Tonix Pharmaceuticals Holding Corp. 16 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO TNX - 601 ER Drug product • The only abuse - deterrent properties approved for the labels of certain marketed opioids are extended - release formulations with physiochemical barriers +/ - aversive components to abuse • TNX - 601 ER was formulated with attention to these potentially abuse deterrent properties: ‒ Active ingredient, tianeptine oxalate less soluble than sodium salt, reducing extraction efficiency in solvents such as water and alcohol ‒ Microcrystalline cellulose is a compression aid that results in extremely hard tablets, reducing ability to crush to fine particulate matter for insufflation or efficient extraction, pressed at >100 Newtons ‒ Inclusion of high molecular weight gel - forming polymers also adversely affects the “syringe - ability” and injectability of the drug product ‒ Inclusion of hydrophilic fumed silica as well as magnesium stearate may cause nasal irritation if insufflated; in high doses, orally ingested magnesium stearate may cause GI hyperactivity and irritation ‒ All potentially serve to make TNX - 601 ER a non - optimal source of tianeptine for high dose abuse TNX - 601 ER formulated with attention to FDA - guided potential abuse deterrent properties* *https://www.fda.gov/drugs/information - drug - class/final - guidance - evaluation - and - labeling - abuse - deterrent - opioids

© 2023 Tonix Pharmaceuticals Holding Corp. 17 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Summary: TNX - 601 ER vs. Other Antidepressants • Given tianeptine’s unique metabolic pathway, which is independent of the hepatic P450 system, it is anticipated that, like tianeptine sodium, TNX - 601 ER will have a reduced risk of drug - drug interactions compared to most antidepressants • Unique mechanism of action (MOA) compared to available antidepressants in the U.S. • The efficacy of tianeptine sodium IR is comparable to both selective serotonin reuptake inhibitor (SSRI) and tricyclic antidepressants 1,2 while being associated with a low incidence of sexual dysfunction compared with either of those classes 3 , and no associated derangement of sleep architecture, sedative effects, weight gain, or cognitive impairment 1 • Once - daily dosing regimen compared to tianeptine sodium IR at three times a day 1 Wagstaff et al., 2001. CNS Drugs . 15(3), 231 – 259 2 Kasper et al., 2002. Eur Psychiatry. 17 (Suppl 3), 331 - 340 3 Bonierbale et al., 2003. Curr Med Res Opin . 19(2):114 - 124

© 2023 Tonix Pharmaceuticals Holding Corp. 18 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO About TNX - 601 ER CNS PORTFOLIO • Tianeptine sodium (amorphous) immediate release (IR) tablets have been available in Europe and many countries in Asia and Latin America for the treatment of MDD since it was first marketed in France in 1989. Due to its short half - life, tianeptine sodium IR is taken three times daily, which is challenging for patient adherence. • Currently, there is no tianeptine - containing product approved in the U.S. and no extended - release tianeptine product approved anywhere in the world. Tonix discovered a novel hemioxalate salt of tianeptine that may provide improved stability, consistency, and manufacturability compared to known salt forms of tianeptine. • TNX - 601 ER is taken once daily, increasing patient adherence and is thereby anticipated to improve the overall effectiveness of treatment compared to that of tianeptine sodium IR. Targeted therapy for Major Depressive Disorder with convenience of once - daily dosing

© 2023 Tonix Pharmaceuticals Holding Corp. 19 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Potential Indications for TNX - 601 ER • Neurodegenerative disorders ‒ Parkinson’s (and associated conditions, e.g. depression and psychosis) 1 ‒ Alzheimer’s (and associated conditions, e.g. agitation, depression and psychosis) 2 • ADHD 3 • Stress disorders 4 ‒ PTSD, Anxiety • Aging/Neuroprotection 5,6 ‒ Mild Cognitive Impairment • Asthma 7 • Overlapping chronic pain syndromes ‒ Fibromyalgia 8 ‒ Irritable bowel syndrome • Addiction ‒ Opiate use disorder 9 ‒ Alcohol use disorder 1 Levin, 2007. Neurosci Behav Physiol . 37(4):419 - 24 2 García - Alberca et al., 2022. J Alzheimers Dis . 88(2):707 - 720 3 Niederhofer et al., 2004. Neuropsychobiology . 49(3): 130 - 3 . 4 Krystal et al., 2009. Drug Discov Today . 14(13 - 14):690 - 697 5 Yoo et al., 2015. J Affect Disord . 185:24 - 30. 6 6 Saiz - Ruiz et al., 1998. Prog. Neuro - Psychopharmacol . & Bio. Psychiat . 22(2): 319 - 329 Informed by clinical data and mechanistic insights 7 Lechin et al., 2004. Methods Find Exp Clin Pharmacol . 26(9): 697 – 701 8 “ ISRCTN16400909 – Tianeptine for the treatment of fibromyalgia: a prospective double - blind, randomised , single - centre , placebo - controlled, parallel group study" . Controlled - trials.com. Archived from the original on 21 July 2010. Retrieved 13 August 2010 9 Chu et al., 2010. Behav Pharmacol . 21(5 - 6):523 - 9

© 2023 Tonix Pharmaceuticals Holding Corp. © 2023 Tonix Pharmaceuticals Holding Corp. THANK YOU