
Tonix Pharmaceuticals Holding Corp. 8-K
Exhibit 99.01

© 2023 Tonix Pharmaceuticals Holding Corp. Corporate Presentation October 2023 NASDAQ: TNXP Version P0496 October 16, 2023 (Doc 1332 )

2 © 2023 Tonix Pharmaceuticals Holding Corp. Cautionary Note on Forward - Looking Statements Certain statements in this presentation regarding strategic plans, expectations and objectives for future operations or results are “forward - looking statements” as defined by the Private Securities Litigation Reform Act of 1995. These statements may be identified by the use of forward - looking words such as “anticipate,” “believe,” “forecast,” “estimate” and “intend,” among others. These forward - looking statements are based on Tonix’s current expectations and actual results could differ materially. There are a number of factors that could cause actual events to differ materially from those indicated by such forward - looking statements. These factors include, but are not limited to, the risks related to failure to obtain FDA clearances or approvals and noncompliance with FDA regulations; risks related to the failure to successfully market any of our products; risks related to the timing and progress of clinical development of our product candidates; our need for additional financing; uncertainties of patent protection and litigation; uncertainties of government or third party payor reimbursement; limited research and development efforts and dependence upon third parties; and substantial competition. As with any pharmaceutical under development, there are significant risks in the development, regulatory approval and commercialization of new products. The forward - looking statements in this presentation are made as of the date of this presentation, even if subsequently made available by Tonix on its website or otherwise. Tonix does not undertake an obligation to update or revise any forward - looking statement, except as required by law. Investors should read the risk factors set forth in the Annual Report on Form 10 - K for the year ended December 31, 2022, as filed with the Securities and Exchange Commission (the “SEC”) on March 13, 2023, and periodic reports and current reports filed with the SEC on or after the date thereof. All of Tonix's forward - looking statements are expressly qualified by all such risk factors and other cautionary statements.

© 2023 Tonix Pharmaceuticals Holding Corp. 3 Who We Are Tonix is committed to developing and marketing therapeutics to treat pain, neurologic, psychiatric and addiction conditions through our central nervous system portfolio and within other areas of high unmet need , including immunology, infectious disease, and rare disease
4 © 2023 Tonix Pharmaceuticals Holding Corp. CNS - Focused Biopharma with Preclinical to Commercial Stage Products Marketed Products For the treatment of acute migraine Robust Development Pipeline Topline data for three late - stage CNS programs expected by end of 2023 Internal Facilities For R&D and clinical - scale manufacturing Strategic Partnerships With world - class academic & research organizations to bring innovative therapeutics to market faster
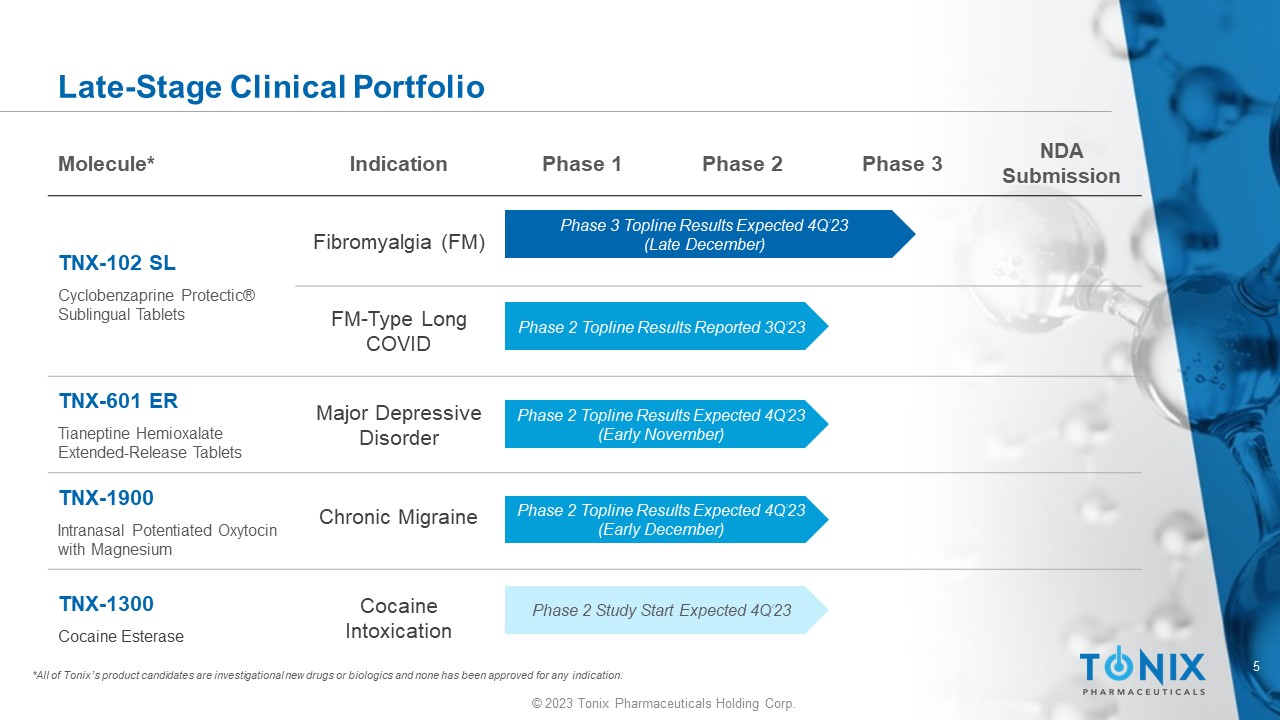
5 © 2023 Tonix Pharmaceuticals Holding Corp. NDA Submission Phase 3 Phase 2 Phase 1 Indication Molecule* Fibromyalgia (FM) TNX - 102 SL Cyclobenzaprine Protectic ® Sublingual Tablets FM - Type Long COVID Major Depressive Disorder TNX - 601 ER Tianeptine Hemioxalate Extended - Release Tablets Chronic Migraine TNX - 1900 Intranasal Potentiated Oxytocin with Magnesium Cocaine Intoxication TNX - 1300 Cocaine Esterase Late - Stage Clinical Portfolio *All of Tonix’s product candidates are investigational new drugs or biologics and none has been approved for any indication. Phase 3 Topline Results Expected 4Q’23 (Late December) Phase 2 Topline Results Reported 3Q’23 Phase 2 Topline Results Expected 4Q’23 (Early December) Phase 2 Topline Results Expected 4Q’23 (Early November) Phase 2 Study Start Expected 4Q’23

© 2023 Tonix Pharmaceuticals Holding Corp. TONIX MEDICINES: MARKETED PRODUCTS

7 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Two Marketed Proprietary Migraine Drugs Non - oral Formulations of Sumatriptan • Each indicated for the tr eatment of acute migraine with or without aura in adults • Sumatriptan remains the acute migraine ‘gold standard’ treatment for many patients and continues to represent the largest segment of the market in terms of unit sales 3 • Each may provide migraine pain relief in as few as 10 minutes for some patients 1,2,4,5 • Patents to 2036 ( Zembrace ) and 2031 ( Tosymra ) 1 Zembrace SymTouch [package insert] . Maple Grove, MN : Upsher - Smith Laboratories, LLC : February 2021 - For more information, talk to your provider and read the Patient Information and Instructions for Use . – Important Safety Information is provided in the appendix 2 Tosymra [package insert]. Maple Grove, MN: Upsher - Smith Laboratories, LLC: Feb 2021. For more information, talk to your provider and read the Patient Information and Instructions for Use. – Important Safety Information is provided in the appendix 3 Upsher - Smith Laboratories, LLC; Data On File, 2023 Zembrace® SymTouch ® (sumatriptan injection) 3 mg 1 Tosymra® (sumatriptan nasal spray) 10 mg 2 Acquired from Upsher - Smith Laboratories which has managed care contracts covering ~200 M lives • Contract includes a transition period during which Tonix expects to secure its own contracts Retail Product Sales for the 12 months ended December 31 st 2022 • Retail sales: ~$23 M (Zembrace ~$19.6 M and Tosymra ~$3.5 M) 4 Tonix is prepared to meet potential increased demand for Tosymra following GSK’s planned d iscontinuation of Imitrex® (sumatriptan) nasal s pray a fter January 2024 4 Mathew NT, et al. Dose ranging efficacy and safety of subcutaneous sumatriptan in the acute treatment of migraine. US Sumatri pta n Research Group. Arch Neurol. 1992;49(12):1271 - 1276. 5 Wendt J, et al. A randomized, double - blind, placebo - controlled trial of the efficacy and tolerability of a 4 - mg dose of subcutan eous sumatriptan for the treatment of acute migraine attacks in adults. Clinical Therapeutics. 2006;28(4):517 - 526. Tonix has contracted to acquire the Zembrace, SymTouch and Tosymra trademarks. Intravail is a registered trademark of Aegis Therapeutics, LLC, a wholly owned subsidiary of Neurelis , Inc.

8 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Zembrace and Tosymra Bypass the GI Tract Bypassing gastrointestinal (GI) tract is potential advantage for treating acute migraine • GI absorption may be inconsistent in migraineurs due to gastric stasis (also called “gastroparesis”) 1 - 4 • Nausea and vomiting are symptoms of migraine 5 which can complicate oral treatment Existing intranasal products • Imitrex® nasal spray (sumatriptan) • Migranal ® (dihydroergotamine) nasal spray – developed by Novartis, sold by Bausch Health New intranasal products bringing attention to non - oral route • Pfizer’s Zavzpret ® ( zavegepant ), FDA approved in March, 2023 1 is the first intranasal gepant • Impel NeuroPharma’s Trudhesa ® (dihydroergotamine) FDA approved 2021 2 1 Pfizer Press Release March 10, 2023. – https://www.pfizer.com/news/press - release/press - release - detail/pfizers - zavzprettm - zavegepant - migraine - nasal - spray 2 Impel Press Release September 3, 2021 - https://impelpharma.com/2021/09/03/impel - neuropharma - announces - u - s - fda - approval - of - trudhesa - dihydroergotamine - mesylate - nasal - spr ay - for - the - acute - treatment - of - migraine/
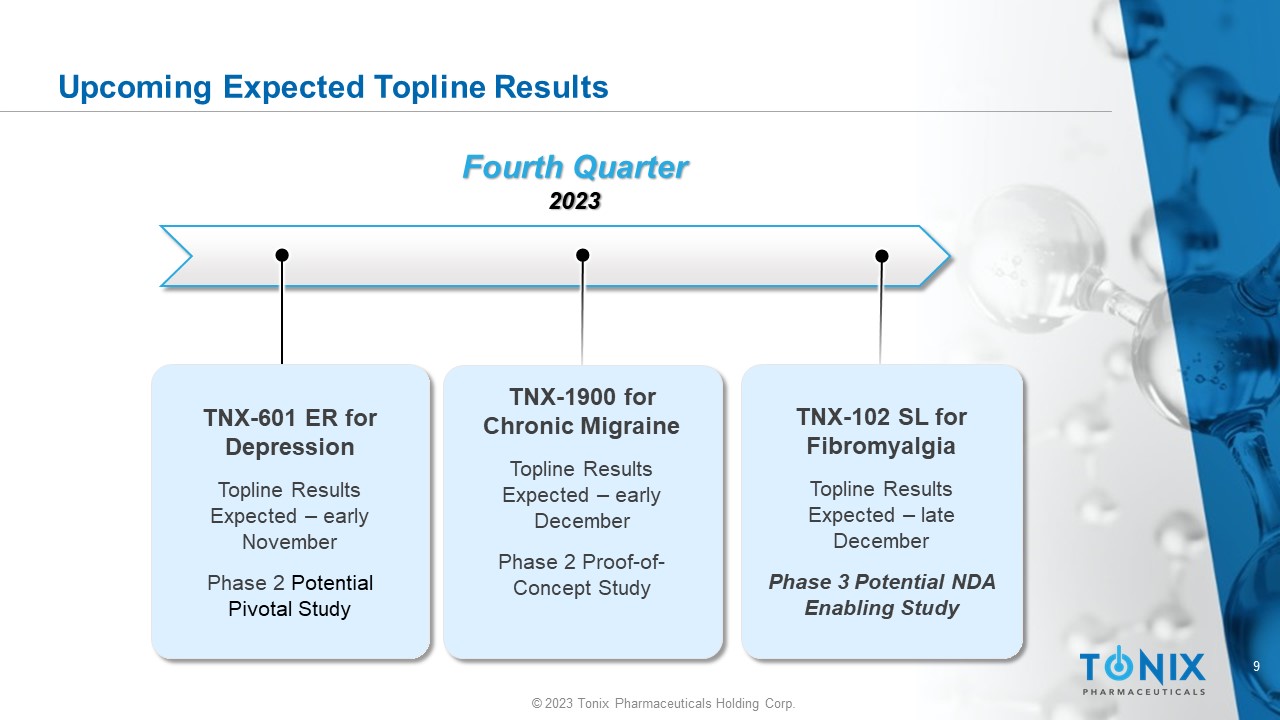
9 © 2023 Tonix Pharmaceuticals Holding Corp. Upcoming Expected Topline Results Fourth Quarter 2023 TNX - 1900 for Chronic Migraine Topline Results Expected – early December Phase 2 Proof - of - Concept Study TNX - 601 ER for Depression Topline Results Expected – early November Phase 2 Potential Pivotal Study TNX - 102 SL for Fibromyalgia T opline Results Expected – late December Phase 3 Potential NDA Enabling Study

© 2023 Tonix Pharmaceuticals Holding Corp. CNS: KEY DEVELOPMENT CANDIDATES

11 © 2023 Tonix Pharmaceuticals Holding Corp. TNX - 102 SL Cyclobenzaprine ( Protectic ® ) A unique, sublingual formulation of cyclobenzaprine designed to optimize delivery and absorption

Unique MOA Facilitates Restorative Sleep: Centrally Acting Analgesic 12 Potent binding and antagonist activities at four key receptors facilitate restorative sleep • serotonergic - 5 - HT2A • adrenergic - α1 • histaminergic - H1 • muscarinic - M1 Relative to Oral Cyclobenzaprine o Lower daytime exposure o Avoids first - pass metabolism o Reduces risk of pharmacological interference from major metabolite Relative to Standard of Care o Potential for better tolerability while maintaining efficacy o Not scheduled nor with recognized abuse potential Key Differentiators © 2023 Tonix Pharmaceuticals Holding Corp. *TNX - 102 SL has not been approved for any indication.

13 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO CNS PORTFOLIO Fibromyalgia a fflicts an estimated 6 - 12 million adults in the US, predominantly women 1 1 American Chronic Pain Association (www.theacpa.org, 2019) 2 Robinson et al, Pain Medicine 2013;14:1400 3 The three drugs with FDA approval for the treatment of fibromyalgia: Pregabalin (Lyrica); Duloxetine (Cymbalta); Milnacipran ( Savella ) 4 Market research by Frost & Sullivan, commissioned by Tonix About Fibromyalgia Fibromyalgia (FM) is a chronic pain disorder resulting from amplified sensory and pain signaling within the CNS. Symptoms include chronic widespread pain, nonrestorative sleep , fatigue, and cognitive dysfunction 6 - 12 million adults Large unmet need: • Patients struggle with daily activities, have impaired quality of life, and frequently are disabled • Physicians and patients report common dissatisfaction with currently marketed products • Average patient has 20 physician office visits per year 2 Current standard of care: • FDA - approved products include Lyrica, Cymbalta, and Savella • Fewer than half of those treated for fibromyalgia receive sustained benefit from the approved drugs 3 • Majority (60%) fail therapy due to lack of a response (25%) or poor tolerability (35%) 4 • Opioid usage is not uncommon

14 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Fibromyalgia Program Status Fibromyalgia TNX - 102 SL Cyclobenzaprine Protectic ® Sublingual Tablets FM - Type Long COVID Phase 3 Topline Results Expected 4Q’23 (Late December) Phase 2 Topline Results Reported 3Q’23 1) One positive Phase 3 study (RELIEF) completed 1 2) Second Phase 3 study (RALLY) missed primary endpoint • Unexpected ~80% increase in adverse event - related discontinuations in both drug and placebo arms, potentially due to recruiting during COVID - 19 3) Confirmatory Phase 3 study (RESILIENT) enrollment complete 1 Lederman et al., (2023) Arthritis Care & Research "Efficacy and Safety of TNX - 102 SL (Sublingual Cyclobenzaprine) for the Treatment of Fibromyalgia: Results From the RELIEF Trial ", doi : 10.1002/acr.25142. Epub ahead of print. PMID: 37165930. *TNX - 102 SL has not been approved for any indication. Next Steps: Potentially confirmatory t opline results expected 4Q 2023 (Late December)

15 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Phase 3 RESILIENT Study Design General s tudy c haracteristics: • Randomized, double - blind, placebo - controlle d study in fibromyalgia • U.S. sites only, completed enrollment of 457 patients Primary Endpoint: • Daily diary pain severity score change from baseline to Week 14 (TNX - 102 SL vs. placebo) • Weekly averages of the daily numerical rating scale scores • Threshold for potential NDA - enabling study is p < 0.05 Key Secondary Endpoints: • Patient Global Impression of Change responder analysis • Fibromyalgia Impact Questionnaire - Revised (FIQ - R) Symptom Domain score • FIQ - R Function Domain score • PROMIS Sleep Disturbance instrument • PROMIS Fatigue instrument • Weekly average of the daily diary assessment of sleep quality Placebo once - daily at bedtime TNX - 102 SL once - daily at bedtime 5.6 mg (2 x 2.8 mg tablets) * * Two week run in at 2.8 mg dose at bedtime, followed by 12 weeks at 5.6 mg dose ClinicalTrials.gov Identifier: NCT05273749 A Phase 3 Study to Evaluate the Efficacy and Safety of TNX - 102 SL Taken Daily in Patients With Fibromyalgia (RESILIENT) 14 weeks

16 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO About Fibromyalgia - Type Long COVID Many Long - COVID symptoms overlap with core symptoms of fibromyalgia and are hallmarks of other chronic pain syndromes like myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) Multisite pain Memory issues Fatigue Sleep disturbances 19% Long COVID occurs in approximately 19% of recovered COVID - 19 patients 2 40 % As many as 40% of Long COVID patients experience multi - site pain 3,4 1 CDC - https://www.cdc.gov/coronavirus/2019 - ncov/long - term - effects/index.html#:~:text=Some%20people%20who%20have%20been,after%20acute%2 0COVID%2D19%20infection . 2 CDC Press Release, June 22, 2022 - https://www.cdc.gov/nchs/pressroom/nchs_press_releases/2022/20220622.htm 3 Harris, H, et al. Tonix data on file. 2022 4 TriNetX Analytics Long COVID is broadly defined as signs, symptoms, and conditions that continue or develop after acute COVID - 19 infection 1

17 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Phase 2 PREVAIL Study Design Study c haracteristics: • Randomized, double - blind, placebo - controlle d study of TNX - 102 SL in fibromyalgia - type Long COVID • U.S. sites only, completed enrollment of 63 patients Primary Endpoint: • Daily diary pain severity score change from baseline to Week 14 (TNX - 102 SL vs. placebo) − Weekly averages of the daily numerical rating scale scores Placebo once - daily at bedtime 14 weeks TNX - 102 SL once - daily at bedtime 5.6 mg (2 x 2.8 mg tablets) * * Two week run in at 2.8 mg dose at bedtime, followed by 12 weeks at 5.6 mg dose ClinicalTrials.gov Identifier: NCT05472090 “A Phase 2 Study to Evaluate the Efficacy and Safety of TNX - 102 SL in Patients With Multi - Site Pain Associated With Post - Acute Sequelae of SARS - CoV - 2 Infection (PREVAIL)” Next Steps: End of Phase 2 Meeting with FDA 1Q 2024

18 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Phase 2 PREVAIL Topline Results 1 D id not meet the primary endpoint of multi - site pain reduction at W eek 14 However, f indings fulfill the objectives of proof - of - concept study, supporting the decision to advance the program b ased on a proposed primary endpoint using the PROMIS Fatigue scale • TNX - 102 SL showed robust effect size in improving fatigue and consistent activity across secondary measures of sleep quality, cognitive function, disability and Patient Global Impression of Change (PGIC) • Was g enerally well tolerated with an adverse event (AE) profile comparable to prior studies with TNX - 102 SL: ‒ AE - related discontinuations were similar in drug and placebo arms ‒ No new safety signals were observed Fatigue is the signature symptom of Long COVID and has been identified as the dominant symptom contributing to disability 2 • W e observed numerical improvement in the PROMIS fatigue score (in RELIEF p= 0.007 MMRM and in RALLY p= 0.007 MMRM) in both prior Phase 3 studies of TNX - 102 SL in fibromyalgia, • W e believe the results of PREVAIL, toge ther with extensive data from studies in other chronic conditions 3 - 5 , makes PROMIS Fatigue a solid candidate for the primary endpoint of future Long COVID registrational studies 1 Tonix Press Release, September 5, 2023 - https://bit.ly/3Z6FQHQ 2 Walker S, et al . BMJ Open 2023;13:e069217. doi:10.1136/ bmjopen - 2022 - 069217 3 Cook, K.F., et al. 2016. Journal of Clinical Epidemiology , 73, 89 - 102 4 Cella, D., et al. 2016. Journal of Clinical Epidemiology , 73, 128 – 134 5 Lai, J.S., et al. 2011. Archives of Physical Medicine and Rehabilitation , 92(10 Supplement), S20 - S27.

19 © 2023 Tonix Pharmaceuticals Holding Corp. TNX - 601 ER Tianeptine Hemioxalate Extended - Release Tablets An innovative approach to treating depression
Improved Formulation of Tianeptine Promotes Neuroplasticity 20 Unique mechanism of action – beyond neurotransmitter modulation Key Differentiators © 2023 Tonix Pharmaceuticals Holding Corp. Relative to tianeptine immediate release available ex - US o Once daily dosing Relative to traditional antidepressants o Tianeptine sodium IR has similar efficacy but less weight gain or sexual side effects than traditional antidepressants o Tianeptine’s side effects are described in labeling in countries in which it is marketed 1 1 Su mmary of product characteristics ( SmPC), European Medicines Agency, Stablon ®. *TNX - 601 ER has not been approved for any indication.
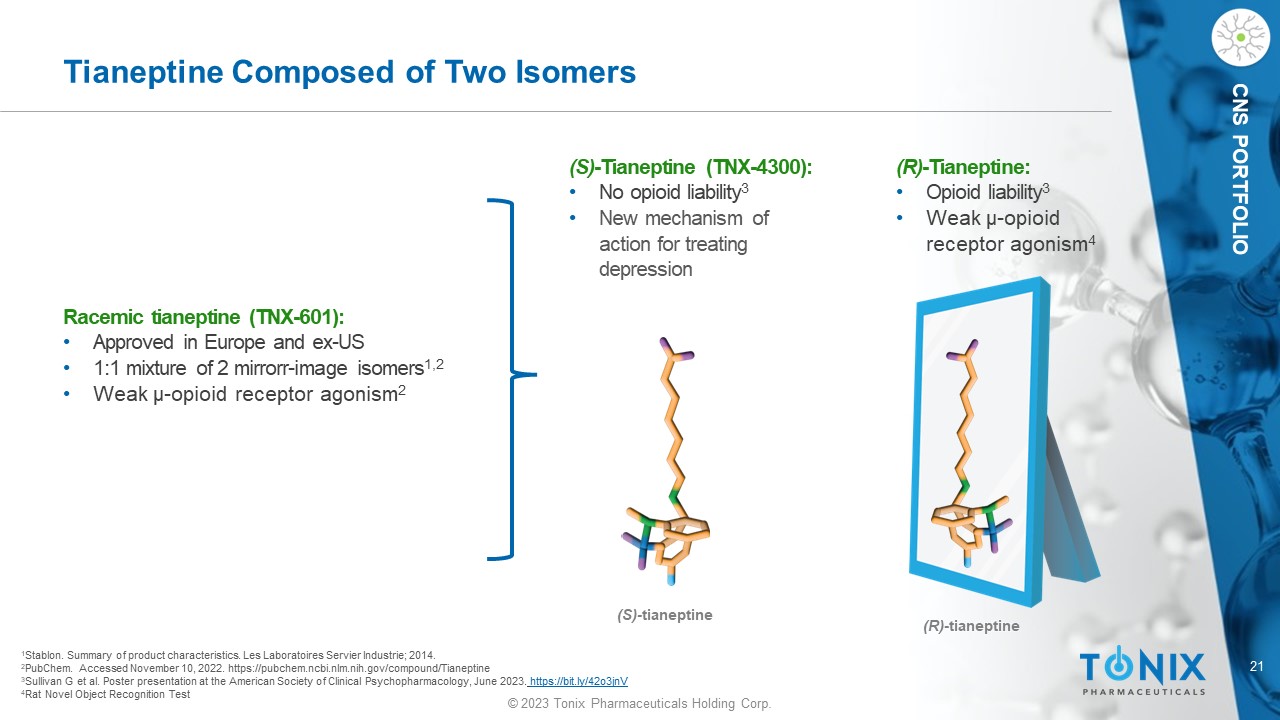
21 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Tianeptine Composed of Two Isomers Racemic tianeptine (TNX - 601) : • Approved in Europe and ex - US • 1:1 mixture of 2 mirror r - image isomers 1,2 • Weak µ - o pioid receptor agonism 2 (S) - t ianeptine (R) - tianeptine 1 Stablon. Summary of product characteristics. Les Laboratoires Servier Industrie ; 2014. 2 PubChem. Accessed November 10, 2022. https://pubchem.ncbi.nlm.nih.gov/compound/Tianeptine 3 Sullivan G et al. Poster presentation at the American Society of Clinical Psychopharmacology, June 2023 . https://bit.ly/42o3jnV 4 Rat Novel Object Recognition Test (S) - Tianeptine (TNX - 4300): • N o opioid liability 3 • New mechanism of action for treating depression (R) - Tianeptine: • Opioid liability 3 • Weak µ - o pioid receptor agonism 4

22 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO CNS PORTFOLIO Depression afflicts nearly 47 million adults in the US (18.4% of population) About Major Depressive Disorder Major depressive disorder (MDD) is a leading cause of disability worldwide, with 21 million adults in the US alone experiencing a depressive episode in 2020 1 . 1 Substance Abuse and Mental Health Services Administration (SAMHSA). 2020. Key Substance Use and Mental Health Indicators in t he United States: Results from the 2020 National Survey on Drug Use and Health. 2 Rush et al., 2004. Control Clin Trials . 25(1):119 - 42 3 CDC - https://www.cdc.gov/mmwr/volumes/72/wr/mm7224a1.htm?s_cid=mm7224a1_w 47 million Current standard of care: • SSRIs are currently the most prescribed class of antidepressants Large unmet need: • O nly about 50% of patients with MDD respond to initial SSRI treatment, and only 35 - 40% of those patients achieve full remission 2 • Antidepressant treatments often continue for years, and the side effect profiles of the monoaminergic antidepressants are intolerable to many

23 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO TNX - 601 ER - Phase 2 UPLIFT* Study Design General s tudy c haracteristics: • Randomized, double - blind, placebo - controlle d study in Major Depressive Disorder to evaluate monotherapy with TNX - 601 ER versus placebo • Parallel design with two arms – treatment with tianeptine hemioxalate 39.4 mg or placebo • U.S. sites only, completed enrollment of 132 patients • Clinical phase completed – announced October 16, 2023 1 Primary Endpoint: • Mean change from baseline in the Montgomery - Åsberg Depression Rating Scale (MADRS) total score at Week 6 • Threshold for achieving positive proof - of - concept study is effect size (ES) > 0.20 • Threshold for positive pivotal study is p - value < 0.05 *ClinicalTrials.gov Identifier: NCT05686408 Abbreviations: Dx, diagnosis; ER, extended - release; F/U, follow - up; MDD, major depressive disorder; MDE, major depressive episo de; N, number 1 Tonix Press Release, October 16 2023: https://ir.tonixpharma.com/news - events/press - releases/detail/1430/tonix - pharmaceuticals - completes - clinical - stage - of - phase - 2 Next Steps: Topline results expected 4Q 2023 (Early November)
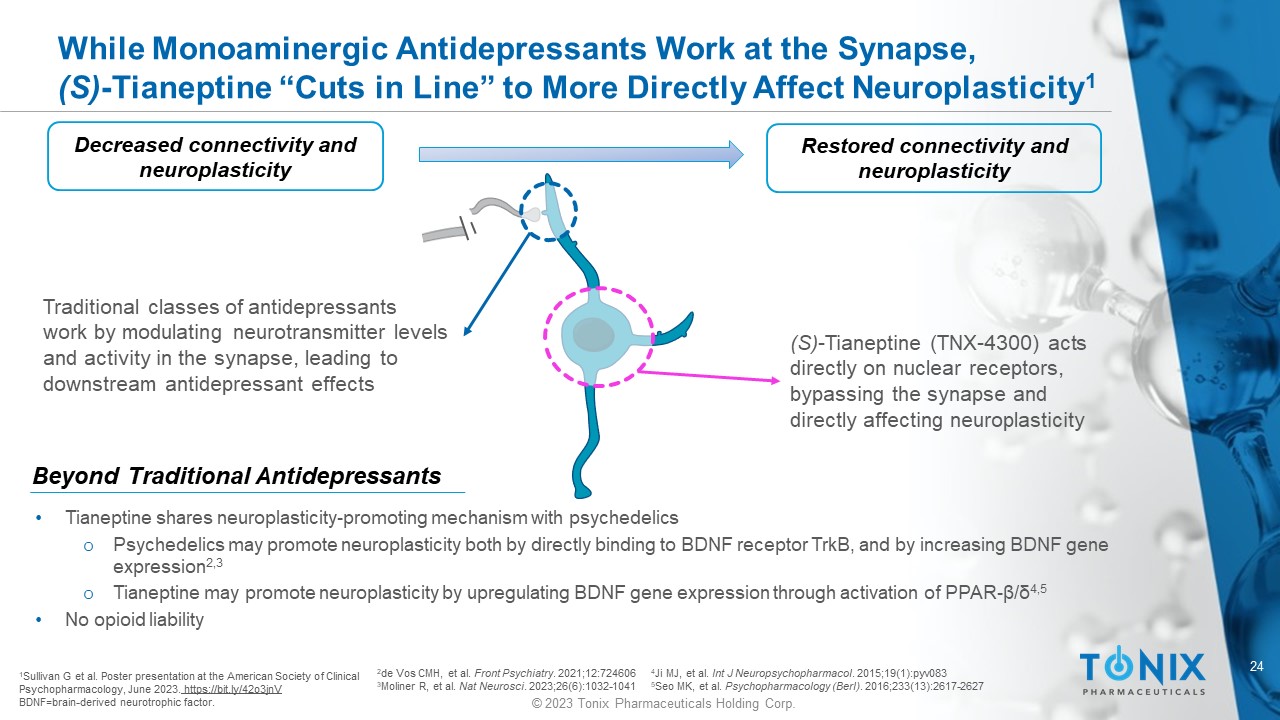
24 © 2023 Tonix Pharmaceuticals Holding Corp. While Monoaminergic Antidepressants Work at the Synapse, (S) - Tianeptine “Cuts in Line” to More Directly Affect Neuroplasticity 1 Decreased connectivity and neuroplasticity 1 Sullivan G et al. Poster presentation at the American Society of Clinical Psychopharmacology, June 2023. https://bit.ly/42o3jnV BDNF=brain - derived neurotrophic factor. Traditional classes of antidepressants work by modulating neurotransmitter levels and activity in the synapse, leading to downstream antidepressant effects (S) - Tianeptine ( TNX - 4300) acts directly on nuclear receptors, bypassing the synapse and directly affecting neuroplasticity Restored connectivity and neuroplasticity • Tianeptine shares neuroplasticity - promoting m echanism w ith p sychedelics o Psychedelics may promote neuroplasticity both by directly binding to BDNF receptor TrkB , and by increasing BDNF gene expression 2 ,3 o Tianeptine may promote neuroplasticity by upregulating BDNF gene expression through activation of PPAR - β/δ 4 ,5 • No opioid liability Beyond Traditional Antidepressants 2 de Vos CMH, et al. Front Psychiatry . 2021;12:724606 3 Moliner R, et al. Nat Neurosci . 2023;26(6):1032 - 1041 4 Ji MJ, et al. Int J Neuropsychopharmacol . 2015;19(1):pyv083 5 Seo MK, et al. Psychopharmacology ( Berl ) . 2016;233(13):2617 - 2627

25 © 2023 Tonix Pharmaceuticals Holding Corp. TNX - 1900 and TNX - 2900 Intranasal Potentiated Oxytocin with Magnesium A n ovel, non - CGRP antagonist approach to treatment
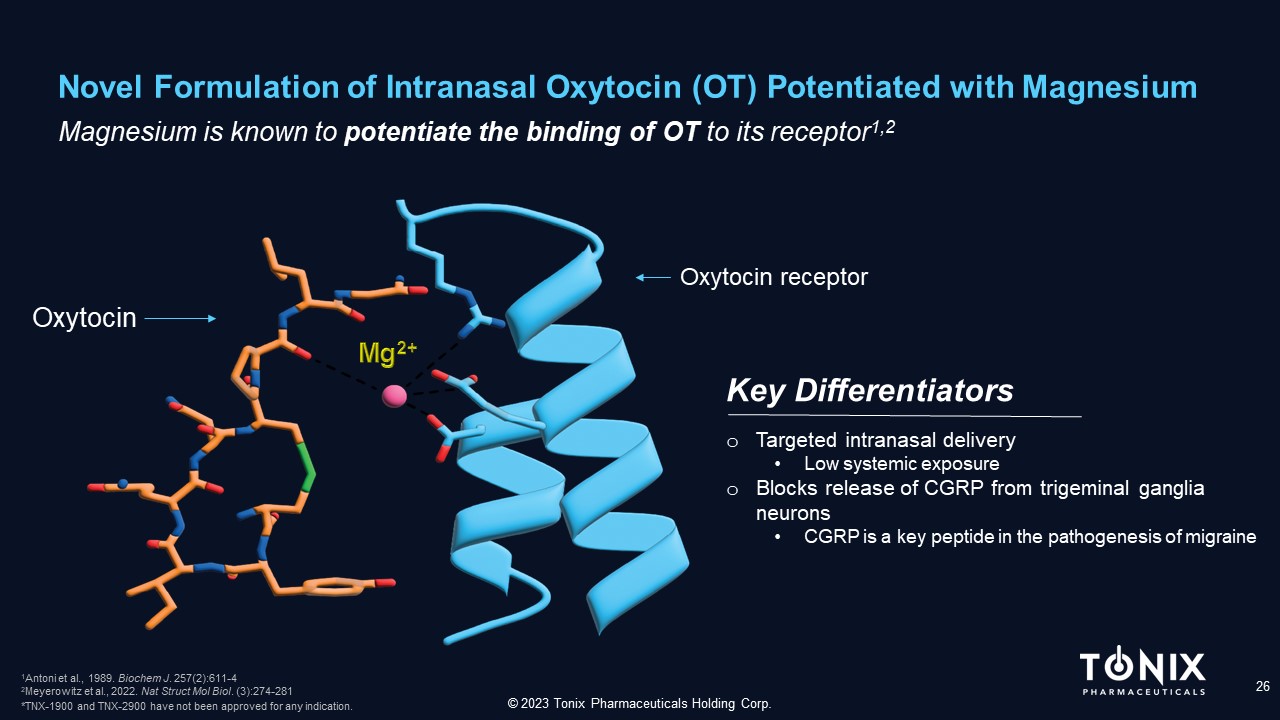
Novel Formulation of Intranasal Oxytocin (OT) Potentiated with Magnesium 26 Magnesium is known to potentiate the binding of OT to its receptor 1 ,2 Key Differentiators © 2023 Tonix Pharmaceuticals Holding Corp. Oxytocin receptor Oxytocin o Targeted intranasal delivery • Low systemic exposure o Blocks release of CGRP from trigeminal ganglia neurons • CGRP is a key peptide in the pathogenesis of migraine 1 Antoni et al., 1989. Biochem J . 257(2):611 - 4 2 Meyerowitz et al., 2022. Nat Struct Mol Biol . (3):274 - 281 *TNX - 1900 and TNX - 2900 have not been approved for any indication.
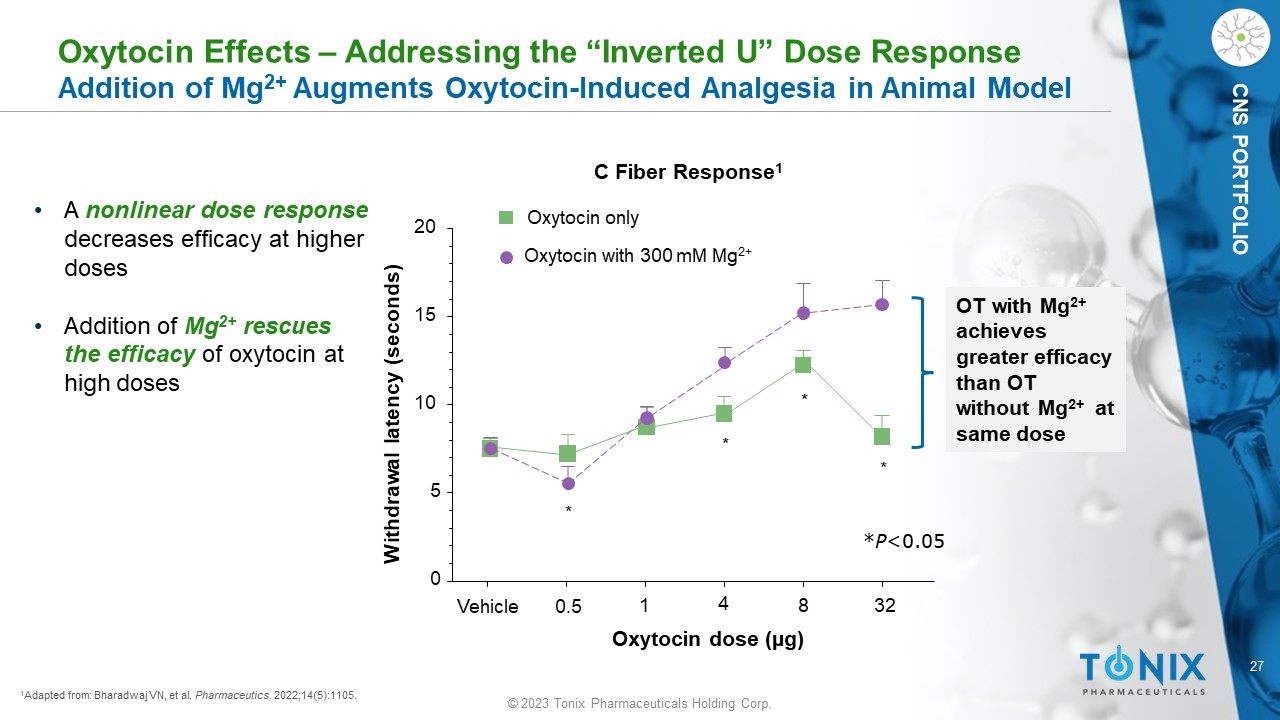
27 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO 1 Adapted from: Bharadwaj VN, et al. Pharmaceutics . 2022;14(5):1105. Vehicle 0.5 1 4 8 32 0 5 10 15 20 Oxytocin only Oxytocin with 300 mM Mg 2+ * * * * Oxytocin dose (µg) Withdrawal latency (seconds) C Fiber Response 1 * P <0.05 Oxytocin Effects – Addressing the “Inverted U” Dose Response Addition of Mg 2+ Augments Oxytocin - Induced Analgesia in Animal Model • A nonlinear dose response decreases efficacy at higher doses • Addition of Mg 2+ rescues the efficacy of oxytocin at high doses OT with Mg 2+ achieves greater efficacy than OT without Mg 2+ at same dose

28 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO CNS PORTFOLIO Chronic migraine afflicts 3 - 7 million adults in the US 1 About Chronic Migraine Chronic migraine involves frequent (>15 days per month) or long - lasting episodes of headaches and migraines over the course of at least 3 months. Migraines can occur with or without an aura and are often debilitating for patients. 3 - 7 million adults 1 Natoli et al., Global prevalence of chronic migraine: a systematic review, Cephalagia , 2010, 30:599 - 609 2 Robbins, At Stake: The Possible Long - Term Side Effects of CGRP Antagonists, https://www.practicalpainmanagement.com/pain/headache/stake - possible - long - term - side - effects - cgrp - antagonists , accessed November 8, 2020. Current standard of care: • Anti - CGRP antibodies and Botox® ( onabotulinumtoxinA ) are specifically approved to prevent headaches in chronic migraine • Nurtec ® (Rimegepant), a gepant , is approved for both prevention of migraine and acute treatment Large unmet need: • Anti - CGRP antibodies and oral gepants involve systemic exposure • Long term safety concerns with prolonged systemic blockade of CGRP receptor 2
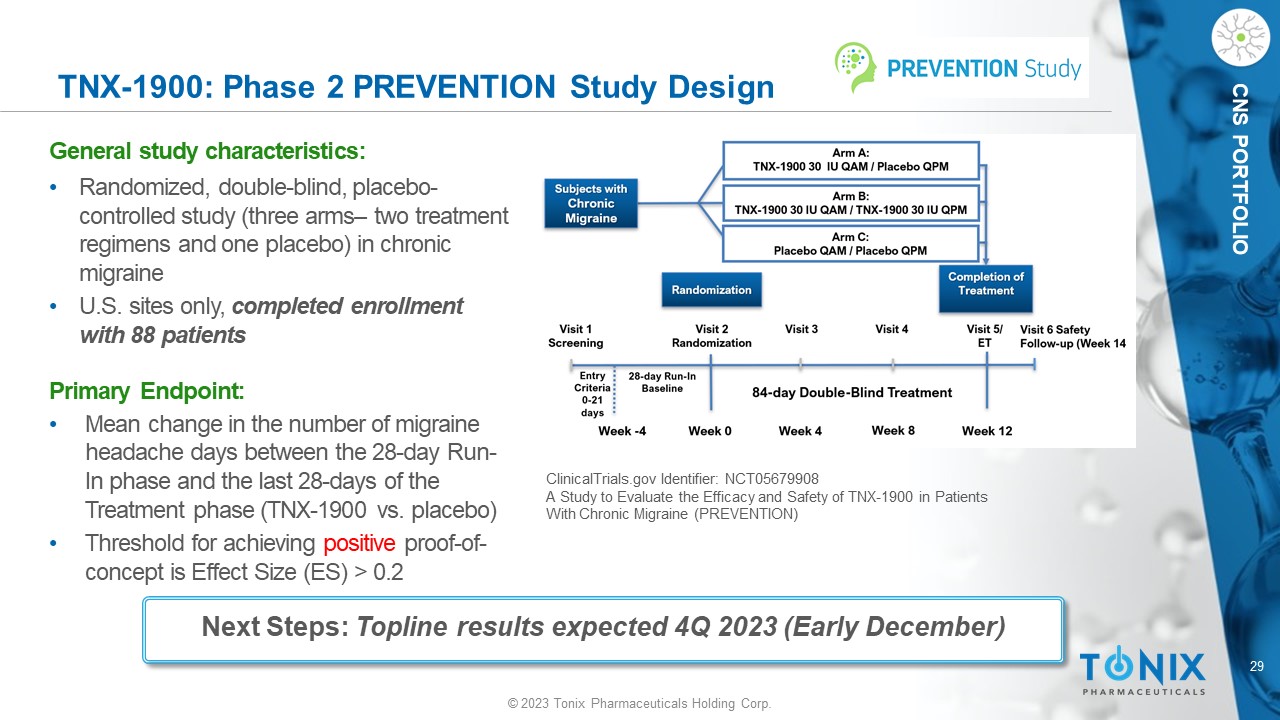
29 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO TNX - 1900: Phase 2 PREVENTION Study Design General s tudy c haracteristics: • Randomized, double - blind, placebo - controlle d study (three arms – two treatment regimens and one placebo) in chronic migraine • U.S. sites only, completed enrollment with 88 patients Primary Endpoint: • M ean change in the number of migraine headache days between the 28 - day Run - In phase and the last 28 - days of the Treatment phase (TNX - 1900 vs. placebo) • Threshold for achieving positive proof - of - concept is Effect Size ( ES) > 0.2 ClinicalTrials.gov Identifier: NCT05679908 A Study to Evaluate the Efficacy and Safety of TNX - 1900 in Patients With Chronic Migraine (PREVENTION) N = 50 N = 50 N = 50 Next Steps: Topline results expected 4Q 2023 (Early December)

30 © 2023 Tonix Pharmaceuticals Holding Corp. Potential Applications of TNX - 1900 & TNX - 2900: Investigator Led Studies Adolescent Obesity Binge Eating Dis. Eating Behavior and Weight Disorders Autism Social Functioning Disorders Social Anxiety Disorder Migraine Craniofacial Pain Pain Conditions Phase 2 Study in Prevention of Headache in Chronic Migraine • Topline results expected 4Q 2023 Phase 2 Biomarker Study Phase 2 Study initiated • Investigator - Initiated IND Phase 2 Study initiated • Investigator - Initiated IND Orphan Drug Designation Awarded • Phase 2 expected to initiate in 2024 Phase 2 Study initiated • Investigator - Initiated IND PWS* *Prader - Willi Syndrome

31 © 2023 Tonix Pharmaceuticals Holding Corp. RARE DISEASE PORTFOLIO Rare genetic diseas e that afflicts 10 - 20 thousand individuals in the US About Prader - Willi Syndrome Prader - Willi Syndrome (PWS) is the most common genetic cause of life - threatening childhood obesity. PWS causes unhealthy behaviors around food 1 - 4 , c onsequences such as obesity, type 2 diabetes, and cardiovascular disease 1 - 5 , and creates significant caretaker burden 1 - 4 10 - 20 thousand individuals *Tonix has been granted FDA Orphan Drug Designation 1 Miller et al., 2011. Am J Med Genet A . 1 55A(5):1040 - 1049 2 Butler et al., 2017. Genet Med. 19(6):635 - 642 3 Butler MG. NORD. Updated 2018. Accessed May 25, 2022. https://rarediseases.org/rare - diseases/prader - willi - syndrome/ 4 Prader - Willi Syndrome Association USA. Accessed May 25, 2022. https://www.pwsausa.org/what - is - prader - willi - syndrome/ 5 Muscogiuri et al., 2021. J Endocrinol Invest . 44(10):2057 - 2070 Current standard of care: • Human growth hormone treatment is FDA - approved for growth failure in PWS children Large unmet need: • Currently no cure, and no treatment for PWS - related hyperphagia • Consequences can be life threatening - obesity and cardiovascular disease are leading cause of death

32 © 2023 Tonix Pharmaceuticals Holding Corp. TNX - 1300 Cocaine Esterase Fast acting antidote for life threatening cocaine intoxication

Recombinant Protein Rapidly Degrades Cocaine in the Bloodstream 33 Drops plasma exposure by 90% in 2 minutes Key Differentiators © 2023 Tonix Pharmaceuticals Holding Corp. o Rapidly metabolizes cocaine within matter of minutes o N o other product currently on the market for this indication CocE Cocaine FDA Breakthrough Therapy Designation Awarded Cooperative Agreement Grant from National Institute on Drug Abuse (NIDA) *TNX - 1300 has not been approved for any indication.

34 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO CNS PORTFOLIO Over 500,000 emergency department visits for cocaine, annually 3,4 About Cocaine Intoxication Over 5 million Americans reported current cocaine use in 2020, which is almost 2% of the population 1 . In 2021, more than 24,900 individuals in the US died from drug overdose deaths involving cocaine 2 500k 1 Substance Abuse and Mental Health Services Administration. (2021). Results from the 2020 National Survey on Drug Use and Health: Detailed Tables: Prevalence Estimates, Standard Errors, and Sample Sizes. 2 Centers for Disease Control and Prevention (CDC) - https://www.cdc.gov/nchs/nvss/vsrr/drug - overdose - data.htm 3 Substance Mental Health Services Administration, Drug Abuse Warning Network, 2011: National Estimates of Drug - Related Emergency Department Visits. HHS Publication No. (SMA) 13 - 4760, DAWN Series D - 39. Rockville, MD: Substance Abuse and Mental Health Services Administration, 2013. 4 Drug Abuse Warning Network, 2011: Selected Tables of National Estimates of Drug - Related Emergency Department Visits. Rockville, MD: Center for Behavioral Health Statistics and Quality, SAMHSA, 2013. Current standard of care: • Patients are currently managed only by supportive care for the adverse effects of cocaine intoxication on the cardiovascular and central nervous systems Large unmet need: • N o other product currently on the market for this indication • TNX - 1300 could significantly reduce the time and resources required for other detox services • Potentially r educes the risk of morbidity and mortality

© 2023 Tonix Pharmaceuticals Holding Corp. IMMUNOLOGY: KEY CANDIDATES

36 © 2023 Tonix Pharmaceuticals Holding Corp. TNX - 1500 Anti - CD40L Monoclonal Antibody Next Generation mAb preserves efficacy without risk of thrombosis

Next Generation anti - CD40L mAb 37 Re - engineered to better modulate the binding of Fc R and mitigate risk of thrombosis Key Differentiators © 2023 Tonix Pharmaceuticals Holding Corp. Selectively Modified anti - CD40L AB Ruplizumab full Fab Contains the full ruplizumab Fab and the engineered Fc region that modulates Fc γ R - binding, while preserving FcRn function Mutated Fc γ R - binding region FcRn - binding region Fc γ R - modulated Fc region First Generation: Development halted due to thromboembolic (TE) complications — blood clots — traced to Fc gamma receptor (Fc R) Second Generation: Eliminated the Fc R TE complication but potency and half life was reduced, limiting utility Third Generation (TNX - 1500): Re - engineered to better modulate the binding of Fc R. Expected to deliver efficacy without compromising safety *TNX - 1500 has not been approved for any indication.

38 © 2023 Tonix Pharmaceuticals Holding Corp. IMMUNOLOGY PORTFOLIO TNX - 1500 Strategy and Status 1 Lassiter, G . , et al . ( 2023 ) . TNX - 1500 , a crystallizable fragment – modified anti - CD 154 antibody, prolongs non - human primate renal allograft survival . American Journal of Transplantation . April 3 , 2023 . https : //doi . org/ 10 . 1016 /j . ajt . 2023 . 03 . 022 2 Miura, S . , et al . ( 2023 ) TNX - 1500 , a crystallizable fragment – modified anti - CD 154 antibody, prolongs non - human primate cardiac allograft survival . American Journal of Transplantation . April 6 , 2023 . https : //doi . org/ 10 . 1016 /j . ajt . 2023 . 03 . 025 Peer reviewed articles: Two articles have recently published in the American Journal of Transplantation that demonstrate TNX - 1500 prolongs non - human primate renal and heart allograft survival. 1,2 Third Indication (and beyond): Autoimmune Diseases ( e.g., Multiple Sclerosis, Sj ö gen’s Syndrome, Systemic Lupus Erythematosus) • These indications require large studies, but represent large target markets Proposed Initial Indication: Prevention of Allograft Rejection Status: Phase 1 currently enrolling • Collaborations ongoing with Mass General Hospital on heart and kidney transplantation in non - human primates Next Steps: Initiate Phase 2 study in Kidney Transplant Recipients 1 2 Second Indication: Hematopoetic Cell Transplant (Bone Marrow Transplant) • Potential to reduce GvHD 3

39 © 2023 Tonix Pharmaceuticals Holding Corp. TNX - 1700 Recombinant Trefoil Factor Family Member 2 (rTFF2 - HSA) Fusion Protein Targeting the toxic tumor micro - environment

Fighting Cancer by Targeting the Tumor Micro - Environment 40 S uppresses myeloid - derived suppressor cells (MDSCs) and activates anti - cancer CD8+ T cells o Different MOA than checkpoint inhibitors o Potential synergy with anti - PD - 1 or anti - PD - L1 monoclonal antibodies Key Differentiators © 2023 Tonix Pharmaceuticals Holding Corp. *TNX - 1700 is in the pre - IND stage of development and has not been approved for any indication. Human Serum Albumin (HSA) TFF2 o mTNX - 1700 (mTFF2 - MSA fusion protein) and anti - PD - 1 monotherapy each was able to evoke anti - tumor immunity in the MC38 model of colorectal cancer 1 o mTNX - 1700 augmented the anti - tumor efficacy of anti - PD - 1 therapy in both the MC38 and the CT26.wt models 1 Prec linical Evidence 1 Daugherty, B. et al. March 6, 2023 Keystone Poster ; https://bit.ly/48nIRHM

41 © 2023 Tonix Pharmaceuticals Holding Corp. IMMUNOLOGY PORTFOLIO People living with colorectal cancer in the US 2 About Gastric and Colorectal Cancer Gastric and colorectal cancer are both leading cancers in the US. C olorectal cancer is the 3 rd leading cause of cancer - related deaths in both men and women. 1 >1.3M Current standard of care: • PD - 1 blockade − However, gastric and colorectal cancer are relatively unresponsive Large unmet need: • Gastric and colorectal cancer have a relative 5 - year survival rate of 35.7% and 65%, respectively − Despite advances in the field, patients are still in need of life saving treatment 1 American Cancer Society, accessed September 2023 - https://www.cancer.org/cancer/types/colon - rectal - cancer/about/key - statistics.html 2 NIH, accessed September 2023 - https://seer.cancer.gov/statfacts/html/colorect.html 3 NIH, accessed September 2023 - https://seer.cancer.gov/statfacts/html/stomach.html >125k People living with gastric cancer in the US 3

© 2023 Tonix Pharmaceuticals Holding Corp. INFECTIOUS DISEASE: KEY CANDIDATES

43 © 2023 Tonix Pharmaceuticals Holding Corp. INFECTIOUS DISEASE PORTFOLIO Internal Development & Manufacturing Capabilities R&D Center (RDC): Frederick, MD • Research advancing CNS and immunology drugs • Accelerated development of vaccines and antiviral drugs against infectious diseases • ~48,000 square feet, BSL - 2 with some areas designated BSL - 3 Advanced Development Center (ADC): North Dartmouth, MA • Development and clinical scale manufacturing of biologics • ~45,000 square feet, BSL - 2

44 © 2023 Tonix Pharmaceuticals Holding Corp. INFECTIOUS DISEASE PORTFOLIO Broad - Spectrum Antiviral Discovery Programs Host - directed antiviral discovery programs CD45 targeted therapeutics • Small molecule therapeutics that reduce endogenous levels of CD45, a protein tyrosine phosphatase • Reduction in CD45 protects against many viruses including the Ebola virus Cathepsin inhibitors • Small molecule therapeutics that inhibit essential cathepsins which are required by viruses such as coronaviruses and filoviruses to infect cells • Activity as monotherapy and in combination with other antivirals Virus - directed antivirals discovery program Viral glycan - targeted engineered biologics • B ind to viral densely branched high - mannose (DBH) glycans • Neutralize circulating virus and stop the entry of the progeny virus into cells • Antiviral activity against a broad range of RNA viruses • Activity as monotherapy and in combination with other antivirals

45 © 2023 Tonix Pharmaceuticals Holding Corp. TNX - 801 Live Virus Vaccine Live virus vaccine platform with multitude of potential applications
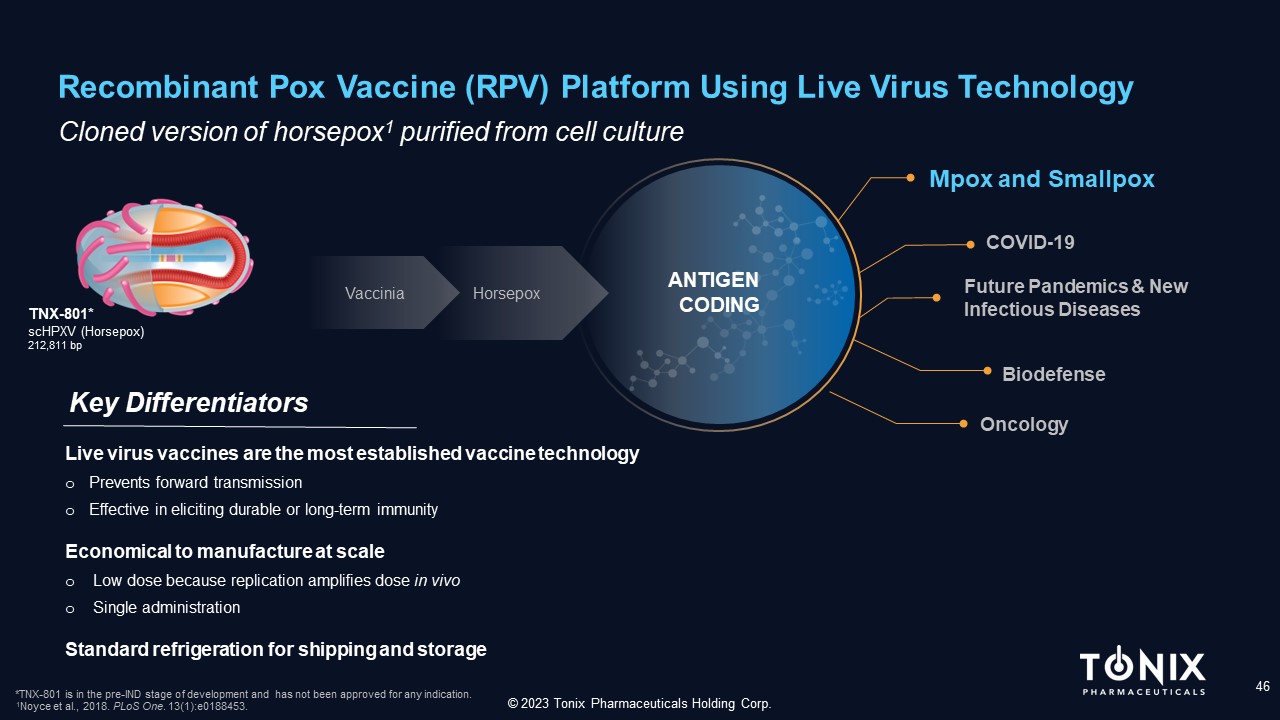
Recombinant Pox Vaccine (RPV) Platform Using Live Virus Technology 46 C loned version of horsepox 1 purified from cell culture Key Differentiators © 2023 Tonix Pharmaceuticals Holding Corp. TNX - 801* scHPXV (Horsepox) 212,811 bp *TNX - 801 is in the pre - IND stage of development and has not been approved for any indication. 1 Noyce et al., 2018. PLoS One . 13(1):e0188453. Live virus vaccines are the most established vaccine technology o Prevents forward transmission o Effective in eliciting durable or long - term immunity Economical to manufacture at scale o Low dose because replication amplifies dose in vivo o Single administration Standard refrigeration for shipping and storage Mpox and Smallpox Future Pandemics & New Infectious Diseases COVID - 19 Biodefense Vaccinia Horsepox Oncology ANTIGEN CODING

© 2023 Tonix Pharmaceuticals Holding Corp. TEAM, NETWORK, & UPCOMING MILESTONES

48 © 2023 Tonix Pharmaceuticals Holding Corp. TNX - 1300: COCAINE INTOXICATION TNX - 1700: GASTRIC AND COLORECTAL CANCERS Key Development Partners TNX - 1500: ALLOGRAFT REJECTION TNX - 1900: MIGRAINE & OTHER INDICATIONS TNX - 801: SMALLPOX AND MONKEYPOX VACCINE TNX - 2900: PRADER - WILLI SYNDROME

49 © 2023 Tonix Pharmaceuticals Holding Corp. Management Team Seth Lederman, MD Co - Founder, CEO & Chairman Jessica Morris Chief Operating Officer Gregory Sullivan, MD Chief Medical Officer Bradley Saenger, CPA Chief Financial Officer

50 © 2023 Tonix Pharmaceuticals Holding Corp. Summary of Upcoming Milestones 4 th Quarter 2023 Clinical Trial Initiations • Phase 2 study of TNX - 1300 for the treatment of cocaine intoxication - expected 4 th Quarter 2023 Data Readouts • Phase 2 UPLIFT study of TNX - 601 ER for major depressive disorder ‒ Affects approximately 47 M adults in the U.S (18.4% of population) 1 • Phase 2 PREVENTION study of TNX - 1900 for chronic migraine ‒ Affects approximately 3 - 7 M adults in the U.S 2 • Phase 3 RESILIENT study of TNX - 102 SL for fibromyalgia ‒ Affects approximately 6 - 12 M adults in the U.S 3 1 CDC - https://www.cdc.gov/mmwr/volumes/72/wr/mm7224a1.htm?s_cid=mm7224a1_w 2 Natoli et al., Global prevalence of chronic migraine: a systematic review, Cephalagia , 2010, 30:599 - 609 3 American Chronic Pain Association (www.theacpa.org, 2019)

© 2023 Tonix Pharmaceuticals Holding Corp. THANK YOU

52 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Zembrace ® Important Safety Information (1 of 2) Zembrace SymTouch ( Zembrace ) can cause serious side effects, including heart attack and other heart problems, which may lead to death. Stop use and get emergency help if you have any signs of a heart attack: D iscomfort in the center of your chest that lasts for more than a few minutes or goes away and comes back; severe tightness, pain, pressure, or heaviness in your chest, throat, neck, or jaw; pain or discomfort in your arms, back, neck, jaw or stomach ; shortness of breath with or without chest discomfort ; breaking out in a cold sweat ; nausea or vomiting ; feeling lightheaded Zembrace is not for people with risk factors for heart disease (high blood pressure or cholesterol, smoking, overweight, diabetes, family history of heart disease) unless a heart exam shows no problem. Do not use Zembrace if you have: H istory of heart problems ; narrowing of blood vessels to your legs, arms, stomach, or kidney (peripheral vascular disease) ; uncontrolled high blood pressure ; hemiplegic or basilar migraines. If you are not sure if you have these, ask your provider. H ad a stroke, transient ischemic attacks (TIAs), or problems with blood circulation ; severe liver problems ; taken any of the following medicines in the last 24 hours: almotriptan , eletriptan , frovatriptan , naratriptan, rizatriptan, ergotamines , dihydroergotamine ; are taking certain antidepressants, known as monoamine oxidase (MAO) - A inhibitors or it has been 2 weeks or less since you stopped taking a MAO - A inhibitor. Ask your provider for a list of these medicines if you are not sure. A n allergy to sumatriptan or any of the components of Zembrace Tell your provider about all of your medical conditions and medicines you take, including vitamins and supplements. Zembrace can cause dizziness, weakness, or drowsiness. If so, do not drive a car, use machinery, or do anything where you need to be alert.

53 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Zembrace ® Important Safety Information (2 of 2) Zembrace may cause serious side effects including: • Changes in color or sensation in your fingers and toes; sudden or severe stomach pain, stomach pain after meals, weight loss, na usea or vomiting, constipation or diarrhea, bloody diarrhea, fever; cramping and pain in your legs or hips; feeling of heaviness or t igh tness in your leg muscles; burning or aching pain in your feet or toes while resting; numbness, tingling, or weakness in your legs; cold fe eli ng or color changes in one or both legs or feet; increased blood pressure including a sudden severe increase even if you have no history of high blood pressure; medication overuse headaches from using migraine medicine for 10 or more days each month. If your headaches g et worse, call your provider. • Serotonin syndrome, a rare but serious problem that can happen in people using Zembrace , especially when used with anti - depressant medicines called SSRIs or SNRIs. Call your provider right away if you have: mental changes such as seeing things that are not th ere (hallucinations), agitation, or coma; fast heartbeat; changes in blood pressure; high body temperature; tight muscles; or tro ubl e walking. • Hives (itchy bumps); swelling of your tongue, mouth, or throat • Seizures even in people who have never had seizures before The most common side effects of Zembrace include: pain and redness at injection site; tingling or numbness in your fingers or toes; dizziness; warm, hot, burning feeling to your face (flushing); discomfort or stiffness in your neck; feeling weak, drowsy, or ti red. Tell your provider if you have any side effect that bothers you or does not go away. These are not all the possible side effe cts of Zembrace . For more information, ask your provider. This is the most important information to know about Zembrace but is not comprehensive. For more information, talk to your provider and read the Patient Information and Instructions for Use . You can also visit www.upsher - smith.com or call 1 - 888 - 650 - 3789. For full Prescribing Information, visit: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=6e5b104f - 2b9e - 416e - 92fb - ef1bdaea867d You are encouraged to report adverse effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1 - 800 - FDA - 1088. Zembrace is a prescription medicine used to treat acute migraine headaches with or without aura in adults who have been diagnosed with migraine. Zembrace is not used to prevent migraines. It is not known if it is safe and effective in children under 18 years of age.

54 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Tosymra® Important Safety Information (1 of 2) Tosymra® can cause serious side effects, including heart attack and other heart problems, which may lead to death. Stop Tosymra and get emergency medical help if you have any signs of heart attack: • D iscomfort in the center of your chest that lasts for more than a few minutes or goes away and comes back ; severe tightness, pain, pressure, or heaviness in your chest, throat, neck, or jaw ; pain or discomfort in your arms, back, neck, jaw, or stomach ; shortness of breath with or without chest discomfort; breaking out in a cold sweat ; nausea or vomiting ; feeling lightheaded Tosymra is not for people with risk factors for heart disease (high blood pressure or cholesterol, smoking, overweight, diabetes, family history of heart disease) unless a heart exam is done and shows no problem. Do not use Tosymra if you have: • H istory of heart problems ; narrowing of blood vessels to your legs, arms, stomach, or kidney (peripheral vascular disease) ; uncontrolled high blood pressure ; severe liver problems ; hemiplegic or basilar migraines. If you are not sure if you have these, ask your healthcare provider. • H ad a stroke, transient ischemic attacks (TIAs), or problems with blood circulation ; taken any of the following medicines in the last 24 hours: almotriptan , eletriptan , frovatriptan , naratriptan, rizatriptan, ergotamines , or dihydroergotamine. Ask your provider if you are not sure if your medicine is listed above • are taking certain antidepressants, known as monoamine oxidase (MAO) - A inhibitors or it has been 2 weeks or less since you stopped taking a MAO - A inhibitor . A sk your provider for a list of these medicines if you are not sure • A n allergy to sumatriptan or any ingredient in Tosymra Tell your provider about all of your medical conditions and medicines you take, including vitamins and supplements. Tosymra can cause dizziness, weakness, or drowsiness. If so, do not drive a car, use machinery, or do anything where you need to be alert.

55 © 2023 Tonix Pharmaceuticals Holding Corp. CNS PORTFOLIO Tosymra may cause serious side effects including: • Changes in color or sensation in your fingers and toes; sudden or severe stomach pain, stomach pain after meals, weight loss, nausea or vomiting, constipation or diarrhea, bloody diarrhea, fever; cramping and pain in your legs or hips, feeling of heaviness or tightness in your leg muscles, burning or aching pain in your feet or toes while resting, numbness, tingling, or weakness in your legs, cold feeling or color changes in one or both legs or feet; increased blood pressure including a sudden severe increase even if you have no history of high blood pressure; medication overuse headaches from using migraine medicine for 10 or more days each month. If your headaches get worse, call your provider . • Serotonin syndrome, a rare but serious problem that can happen in people using Tosymra, especially when used with anti - depressant medicines called SSRIs or SNRIs. Call your provider right away if you have : mental changes such as seeing things that are not there (hallucinations), agitation, or coma; fast heartbeat; changes in blood pressure; high body temperature; tight muscles; or trouble walking. • Seizures even in people who have never had seizures before The most common side effects of Tosymra include : tingling, dizziness, feeling warm or hot, burning feeling, feeling of heaviness, feeling of pressure, flushing, feeling of tightness, numbness, application site (nasal) reactions, abnormal taste, and throat irritation. Tell your provider if you have any side effect that bothers you or does not go away. These are not all the possible side effects of Tosymra. For more information, ask your provider. This is the most important information to know about Tosymra but is not comprehensive. For more information, talk to your provider and read the Patient Information and Instructions for Use . You can also visit www.upsher - smith.com or call 1 - 888 - 650 - 3789. For full Prescribing Information, visit: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=015a5cf9 - f246 - 48bc - b91e - cd730a53d8aa You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch , or call 1 - 800 - FDA - 1088. Tosymra is a prescription medicine used to treat acute migraine headaches with or without aura in adults. Tosymra is not used to treat other types of headaches such as hemiplegic or basilar migraines or cluster headaches. Tosymra is not used to prevent migraines. It is not known if Tosymra is safe and effective in children under 18 years of age. Tosymra ® Important Safety Information (2 of 2)