
Exhibit 99.1

© 2017 Tonix Pharmaceuticals Holding Corp. July 2017 Version P0072 7 - 6 - 17 Investor Presentation

© 2017 Tonix Pharmaceuticals Holding Corp. 2 Cautionary Note on Forward - Looking Statements Certain statements in this presentation regarding strategic plans, expectations and objectives for future operations or results are “forward - looking statements” as defined by the Private Securities Litigation Reform Act of 1995 . These statements may be identified by the use of forward - looking words such as “anticipate,” “believe,” “forecast,” “estimate” and “intend,” among others . These forward - looking statements are based on Tonix’s current expectations and actual results could differ materially . There are a number of factors that could cause actual events to differ materially from those indicated by such forward - looking statements . These factors include, but are not limited to, substantial competition ; our need for additional financing ; uncertainties of patent protection and litigation ; uncertainties of government or third party payor reimbursement ; limited research and development efforts and dependence upon third parties ; and risks related to failure to obtain U . S . Food and Drug Administration clearances or approvals and noncompliance with its regulations . As with any pharmaceutical under development, there are significant risks in the development, regulatory approval and commercialization of new products . The forward - looking statements in this presentation are made as of the date of this presentation, even if subsequently made available by Tonix on its website or otherwise . Tonix does not undertake an obligation to update or revise any forward - looking statement, except as required by law . Investors should read the risk factors set forth in the Annual Report on Form 10 - K for the year ended December 31 , 2016 , as filed with the Securities and Exchange Commission (the “SEC”) on April 13 , 2017 , and future periodic reports filed with the SEC on or after the date hereof . All of Tonix's forward - looking statements are expressly qualified by all such risk factors and other cautionary statements .

© 2017 Tonix Pharmaceuticals Holding Corp. 3 Tonmya ® (Cyclobenzaprine HCl Sublingual Tablets) for Posttraumatic Stress Disorder (PTSD) Phase 3 HONOR study of Tonmya 1 in military - related PTSD enrolling • Encouraging evidence of safety and efficacy was demonstrated in Phase 2 Breakthrough Therapy designation from FDA • Expedited development and accelerated review are expected • Potential to file NDA 2 based on one Phase 3 study if data are statistically persuasive Proposed registration plan agreed by the FDA • Additional nonclinical safety and clinical abuse potential studies are not required Patent protection through 2034 in U.S. 3 • Composition of matter patent for eutectic, required for transmucosal delivery of cyclobenzaprine Novel mechanism targets sleep quality • Memory processing during sleep is important to recovery 1 Tonmya (cyclobenzaprine HCl sublingual tablets) is an investigational new drug and has not been approved for any indication. Tonmya is the proposed proprietary name for TNX - 102 SL for the treatment of PTSD. Tonmya has received U.S. Food and Drug Administration (FDA) conditional acceptance. 2 New Drug Application 3 U.S. Patent No. 9,636,408 for eutectic proprietary Protectic ™ formulation

© 2017 Tonix Pharmaceuticals Holding Corp. 4 Phase 3 HONOR Study in PTSD Enrolling • General s tudy c haracteristics: • Randomized, double - blind, placebo - controlled, entrance CAPS - 5 ≥ 33 • One unblinded interim analys i s (IA) by an independent data monitoring committee at 50% randomized • IA (N ~ 275 ) for efficacy stop , continuation as planned or sample size adjustment • Potential to enroll 550 participants • Approximately 3 5 U.S. clinical sites • Primary e fficacy e ndpoint: • Mean change from baseline in total CAPS - 5 at Week 12 compared between Tonmya 5.6 mg and placebo Placebo once - daily at bedtime 12 weeks Tonmya once - daily at bedtime N ~ 275 (140*) N ~ 275 (140*) 5.6 mg 1H 2018 - IA outcome anticipated 2H 2018 – topline data anticipated, if 550 participants are studied • To confirm Phase 2 AtEase findings in military - related PTSD: • Larger adaptive design study • Enrollment started in 1Q 2017 * Interim analysis open - label extension

© 2017 Tonix Pharmaceuticals Holding Corp. 5 Breakthrough Therapy Designation • FDA granted Tonmya Breakthrough Therapy designation – reported December 19, 2016 • PTSD is a serious condition • Tonmya has potential advantages over existing therapies in military - related PTSD • Benefits of Breakthrough Therapy designation • Eligibility for priority review of the NDA within 6 months instead of 10 months • Option to submit completed portions of the NDA for rolling review • An organizational commitment involving FDA's senior managers to accelerate the development and approval process, an opportunity to compress development time • NDA filing based on HONOR study is possible if results are statistically persuasive • Discussed at March 9, 2017 Initial Cross - disciplinary Breakthrough Meeting with the FDA

© 2017 Tonix Pharmaceuticals Holding Corp. 6 No Recognized Abuse Potential in Clinical Studies • Active ingredient is cyclobenzaprine, which is structurally related to tricyclic antidepressants • Cyclobenzaprine interacts with receptors that regulate sleep quality: 5 - HT 2a ; α 1 - adrenergic and histamine H 1 receptors • Cyclobenzaprine does NOT interact with the same receptors as traditional hypnotic sleep drugs, benzodiazepines or non - benzodiazepines that are associated with retrograde amnesia • Cyclobenzaprine - containing product was approved 40 years ago and current labeling (May 2016) indicates no abuse and dependence concern • Tonmya NDA can be filed without abuse assessment studies • Discussed at March 9, 2017 Initial Cross - disciplinary Breakthrough Meeting with the FDA

© 2017 Tonix Pharmaceuticals Holding Corp. 7 Tonmya Intellectual Property – U.S. Protection until 2034 Composition of matter (eutectic) • U.S. Patent No. 9,636,408 issued May 2, 2017 by U.S. Patent and Trademark Office • Protection expected to 2034 • Additional claims and jurisdictions pending Pharmacokinetics (PK) • Patents filed • Protection expected to 2033 Method of use • Patents filed
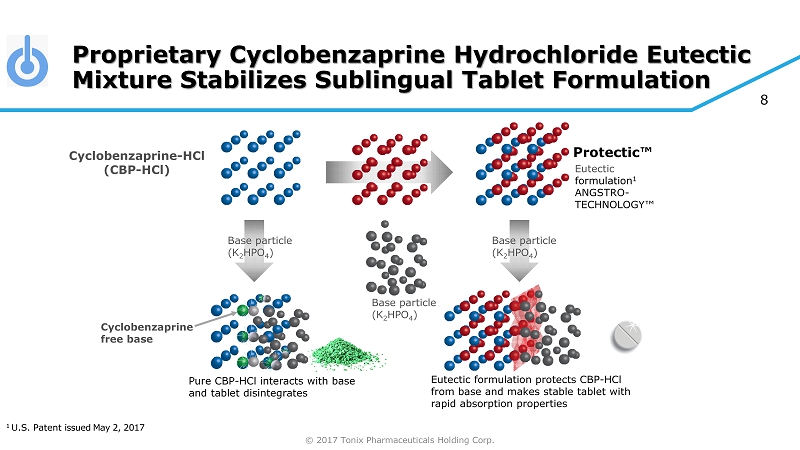
© 2017 Tonix Pharmaceuticals Holding Corp. 8 Proprietary Cyclobenzaprine Hydrochloride Eutectic Mixture Stabilizes Sublingual Tablet Formulation Base particle (K 2 HPO 4 ) Base particle (K 2 HPO 4 ) Base particle (K 2 HPO 4 ) Eutectic formulation 1 ANGSTRO - TECHNOLOGY™ Cyclobenzaprine - HCl (CBP - HCl) Eutectic formulation protects CBP - HCl from base and makes stable tablet with rapid absorption properties Pure CBP - HCl interacts with base and tablet disintegrates Cyclobenzaprine free base Protectic™ 1 U.S. Patent issued May 2, 2017

© 2017 Tonix Pharmaceuticals Holding Corp. 9 Tonmya : Sublingual Formulation is Designed for Bedtime Administration Tonmya : Proprietary sublingual formulation of CBP with transmucosal absorption • Innovation by design with patent protected CBP and mannitol eutectic formulation • Rapid systemic exposure • Increases bioavailability during sleep • Avoids first - pass metabolism • Lowers exposure to long - lived active major metabolite, norcyclobenzaprine ( norCBP ) CBP undergoes extensive first - pass hepatic metabolism when orally ingested • Active major metabolite, norCBP 1 • Long half - life (~72 hours) • Less selective for target receptors ( 5 - HT 2A, a 1 - adrenergic, histamine H 1 ) • More selective for norepinephrine transporter 1 Daugherty et al., Abstract 728, Society of Biological Psychiatry 70th Annual Scientific Convention, May 14 - 16, 2015, Toronto Ont ario, Canada

© 2017 Tonix Pharmaceuticals Holding Corp. 10 Tonmya : Novel Mechanism Targets Sleep Quality for Recovery from PTSD PTSD is a disorder of recovery • Most people exposed to extreme trauma recover over a few weeks • In PTSD, recovery process impeded due to insufficient sleep - dependent memory processing Memory processing is essential to recovery • Vulnerability to memory intrusions and trauma triggers remains if no consolidation of new learning (extinction) Tonmya targets sleep quality 1 • Cyclobenzaprine interacts with receptors that regulate sleep quality: strongly binds and potently blocks 5 - HT 2A , a 1 - adrenergic and histamine H 1 receptors, permissive to sleep - dependent recovery processes 1 Daugherty et al., Abstract 728, Society of Biological Psychiatry 70th Annual Scientific Convention, May 14 - 16, 2015, Toronto Ont ario, Canada

© 2017 Tonix Pharmaceuticals Holding Corp. 11 What are the Symptoms of PTSD? Symptoms of PTSD fall into four clusters: 1. Intrusion (aversive memories, nightmares, flashbacks) 2. Avoidance (avoiding persons, places or situations) 3. Mood/cognitions (memory block, emotional numbing, detachment from others) 4. Hyperarousal (anxiety, agitation & sleep disturbance) Clinician Administered PTSD Scale (CAPS - 5) is used to assess symptom severity and treatment effect • Recognized as the standard for rating PTSD severity in clinical trials • Takes into account all four symptom clusters

© 2017 Tonix Pharmaceuticals Holding Corp. 12 What are the Consequences of PTSD? Consequences: • Impaired daily function and substantial interference with work and social interactions • Reckless or destructive behavior • Increased health care utilization and greater medical morbidity PTSD as a risk factor for: • Depression • Alcohol or substance abuse • Absenteeism/unemployment • Homelessness • Violent acts • Suicidal thoughts and suicide

© 2017 Tonix Pharmaceuticals Holding Corp. 13 PTSD U.S. Prevalence and Index Traumas A chronic response to traumatic event(s) • A majority of people will experience a traumatic event at some point in their lifetime 1 • 20% of women and 8% of men in the U.S. who experience significant trauma develop PTSD 1 • Lifetime prevalence: 6.8% 2 (~ 17 million adults in the U.S.) • Persistent - >1/3 fail to recover, even after several years following the trauma 2 • Twelve month prevalence: U.S. 3.5% 3 (~ 8.6 million adults) EU 2.3% 4 (~10 million adults) Most common forms of trauma 1 • Witnessing someone being badly injured or killed • Natural disaster • Life - threatening accident • Sexual or physical assault 1 Kessler et al, Arch Gen Psychiatry 1995; 52:1048 2 Kessler et al., Arch Gen Psychiatry. 2005; 62:593 3 Kessler et al., Arch Gen Psychiatry. 2005 ; 62:617 ; Prevalence rate of 3.5% applied to U.S. Census estimate of 247 million U.S. adult ( > 18) population in 2015 (www.census.gov/quickfacts/table/PST045215/00) 4 The European Union Market Potential for a New PTSD Drug. Prepared for Tonix Pharmaceuticals by Procela Consultants Ltd Septe mbe r 2016
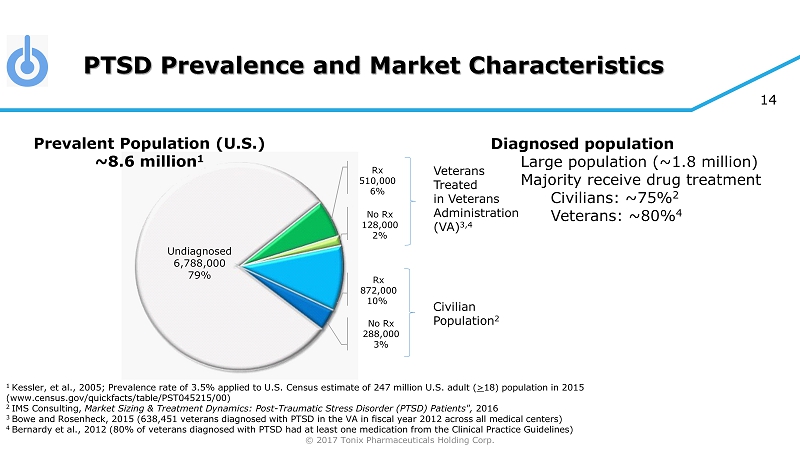
© 2017 Tonix Pharmaceuticals Holding Corp. 14 PTSD Prevalence and Market Characteristics 1 Kessler, et al., 2005; Prevalence rate of 3.5% applied to U.S. Census estimate of 247 million U.S. adult ( > 18) population in 2015 (www.census.gov/quickfacts/table/PST045215/00) 2 IMS Consulting, Market Sizing & Treatment Dynamics: Post - Traumatic Stress Disorder (PTSD) Patients", 2016 3 Bowe and Rosenheck , 2015 (638,451 veterans diagnosed with PTSD in the VA in fiscal year 2012 across all medical centers) 4 Bernardy et al., 2012 (80% of veterans diagnosed with PTSD had at least one medication from the Clinical Practice Guidelines) Veterans Treated in Veterans Administration (VA) 3,4 Civilian Population 2 Diagnosed population Large population (~1.8 million) Majority receive drug treatment Civilians: ~75% 2 Veterans: ~80% 4 Undiagnosed 6,788,000 79% Prevalent Population (U.S.) ~8.6 million 1 Rx 510,000 6% No Rx 128,000 2% Rx 872,000 10% No Rx 288,000 3%
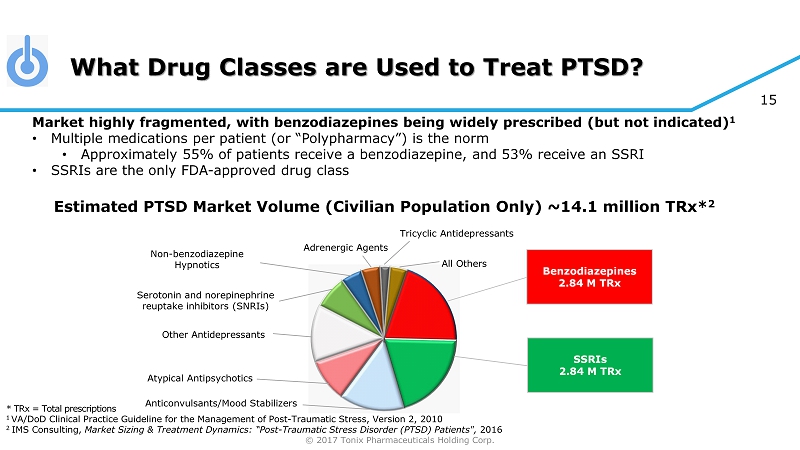
© 2017 Tonix Pharmaceuticals Holding Corp. 15 What Drug Classes are Used to Treat PTSD? * TRx = Total prescriptions 1 VA/DoD Clinical Practice Guideline for the Management of Post - Traumatic Stress, Version 2, 2010 2 IMS Consulting, Market Sizing & Treatment Dynamics: “Post - Traumatic Stress Disorder (PTSD) Patients", 2016 Benzodiazepines 2.84 M TRx SSRIs 2.84 M TRx Other Antidepressants Non - benzodiazepine Hypnotics Serotonin and norepinephrine reuptake inhibitors (SNRIs) Tricyclic Antidepressants Adrenergic Agents All Others Atypical Antipsychotics Anticonvulsants/Mood Stabilizers Market highly fragmented, with benzodiazepines being widely prescribed (but not indicated) 1 • Multiple medications per patient (or “Polypharmacy”) is the norm • Approximately 55% of patients receive a benzodiazepine, and 53% receive an SSRI • SSRIs are the only FDA - approved drug class Estimated PTSD Market Volume (Civilian Population Only) ~14.1 million TRx * 2

© 2017 Tonix Pharmaceuticals Holding Corp. 16 PTSD: Not Well - Served by Approved Treatments FDA approved selective serotonin reuptake inhibitors (SSRIs), paroxetine and sertraline, have not shown efficacy in military - related PTSD Majority of patients unresponsive or intolerant to current treatments • Side effects relating to sexual dysfunction (particularly in males) and sleep are commonly reported Drug therapy compatible and complementary with behavioral therapy • Lack of retrograde amnesia (e.g., off - label use benzodiazepines and non - benzodiazepines) • Lack of interference on sleep (e.g., approved SSRIs)

© 2017 Tonix Pharmaceuticals Holding Corp. 17 Why Initially Target Military - Related PTSD? 1 Friedman et al., J Clin Psychiatry 2007; 68:711 2 Zoloft Package Insert, August, 2014 3 Paxil Package Insert, June, 2014 4 Fava et al., Psychother Psychosom 84:72 - 81, 2015 Military - related PTSD not well - served by existing FDA - approved therapies • No clear treatment response observed in U.S. military population Sertraline: failed to show efficacy in a large multicenter trial in U.S. military (placebo numerically better) 1 Paroxetine: no large trials conducted with predominantly military trauma • Inconsistent treatment response observed in males Sertraline: FDA - conducted post - hoc analysis concluded no effect for male civilian subgroup 2 Paroxetine: no sex - related difference in treatment outcomes 3 • Important tolerability issues with SSRIs in this population Sexual dysfunction 2,3 Insomnia 2,3 SSRI withdrawal syndrome 4
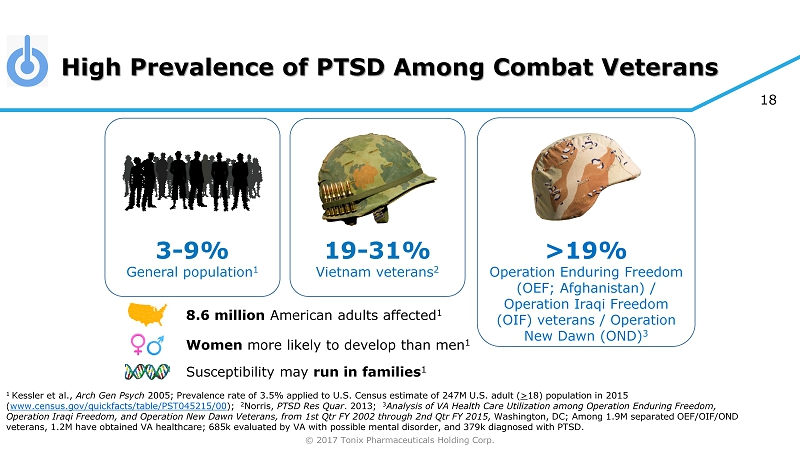
© 2017 Tonix Pharmaceuticals Holding Corp. 18 High Prevalence of PTSD Among Combat Veterans 1 Kessler et al., Arch Gen Psych 2005; Prevalence rate of 3.5% applied to U.S. Census estimate of 247M U.S. adult ( > 18) population in 2015 ( www.census.gov/quickfacts/table/PST045215/00 ) ; 2 Norris, PTSD Res Quar . 2013; 3 Analysis of VA Health Care Utilization among Operation Enduring Freedom, Operation Iraqi Freedom, and Operation New Dawn Veterans, from 1st Qtr FY 2002 through 2nd Qtr FY 2015, Washington, DC ; Among 1.9M separated OEF/OIF/OND veterans, 1.2M have obtained VA healthcare; 685k evaluated by VA with possible mental disorder, and 379k diagnosed with PTSD. >19% Operation Enduring Freedom (OEF; Afghanistan) / Operation Iraqi Freedom (OIF) veterans / Operation New Dawn (OND) 3 3 - 9% General population 1 19 - 31% Vietnam veterans 2 8.6 million American adults affected 1 Women more likely to develop than men 1 Susceptibility may run in families 1

© 2017 Tonix Pharmaceuticals Holding Corp. 19 Growing Economic and Social Burden to Care for Veterans with PTSD Health care costs associated with PTSD for OEF/OIF/OND veterans: Direct costs Indirect costs 1 CBO Report 2012; 2 Tanielan , Invisible Wounds of War . 2005; 3 Analysis of VA Health Care Utilization among Operation Enduring Freedom, Operation Iraqi Freedom, and Operation New Dawn Veterans, from 1st Qtr FY 2002 through 2nd Qtr FY 2015, Washington, DC ; OEF/OIF/OND, Operations Enduring Freedom, Iraqi Freedom and New Dawn. $3,000 - 5,000 per patient per year for OEF/OIF Veterans 1 ~ 1.9M Veterans out of 2.7M Service members deployed between 10/1/2001 and 3/31/2015 3 $2 - 3 billion estimated yearly cost to society 2 Families, social care agencies, schools, employers, welfare system 2
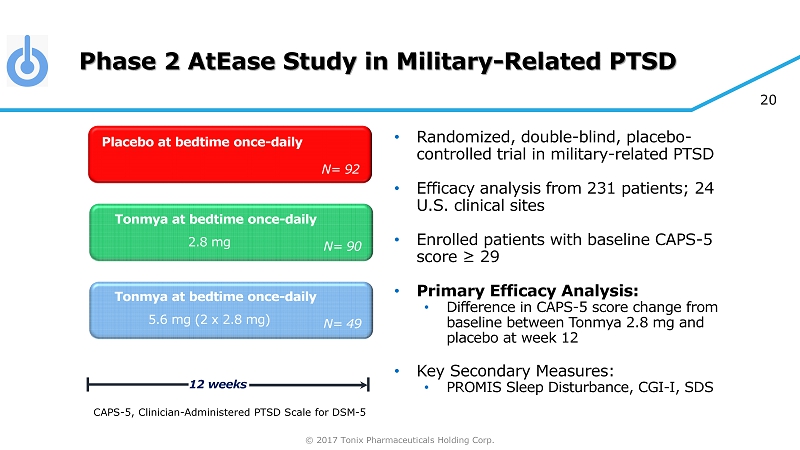
© 2017 Tonix Pharmaceuticals Holding Corp. 20 Phase 2 AtEase Study in Military - Related PTSD • Randomized, double - blind, placebo - controlled trial in military - related PTSD • Efficacy analysis from 231 patients; 24 U.S. clinical sites • Enrolled patients with baseline CAPS - 5 score ≥ 29 • Primary Efficacy Analysis: • Difference in CAPS - 5 score change from baseline between Tonmya 2.8 mg and placebo at week 12 • Key Secondary Measures: • PROMIS Sleep Disturbance, CGI - I, SDS Tonmya at bedtime once - daily Placebo at bedtime once - daily 12 weeks N= 90 Tonmya at bedtime once - daily N= 92 N= 49 2.8 mg 5.6 mg (2 x 2.8 mg) CAPS - 5, Clinician - Administered PTSD Scale for DSM - 5

© 2017 Tonix Pharmaceuticals Holding Corp. 21 Results of Phase 2 AtEase Study in Military - Related PTSD Tonmya 5.6 mg showed clinical benefit in military - related PTSD • CAPS - 5 scale, was statistically significant by Mixed effect Model Repeated Measures, or MMRM, with Multiple Imputation, or MI, analysis (p - value = 0.031) • Dose - effect on multiple efficacy and safety measurements in the AtEase study Well tolerated • No serious adverse events (AE) related to treatment • The most common AEs were local site - administration reactions, including mild and transient tongue numbness
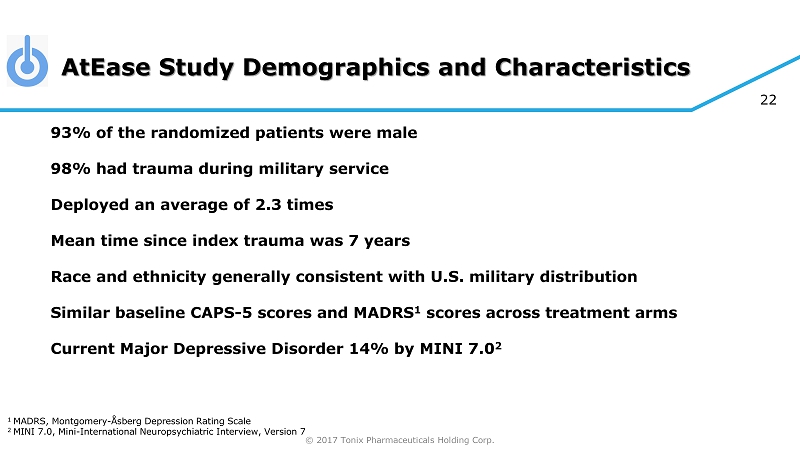
© 2017 Tonix Pharmaceuticals Holding Corp. 22 AtEase Study Demographics and Characteristics 93% of the randomized patients were male 98% had trauma during military service Deployed an average of 2.3 times Mean time since index trauma was 7 years Race and ethnicity generally consistent with U.S. military distribution Similar baseline CAPS - 5 scores and MADRS 1 scores across treatment arms Current Major Depressive Disorder 14% by MINI 7.0 2 1 MADRS, Montgomery - Åsberg Depression Rating Scale 2 MINI 7.0, Mini - International Neuropsychiatric Interview, Version 7
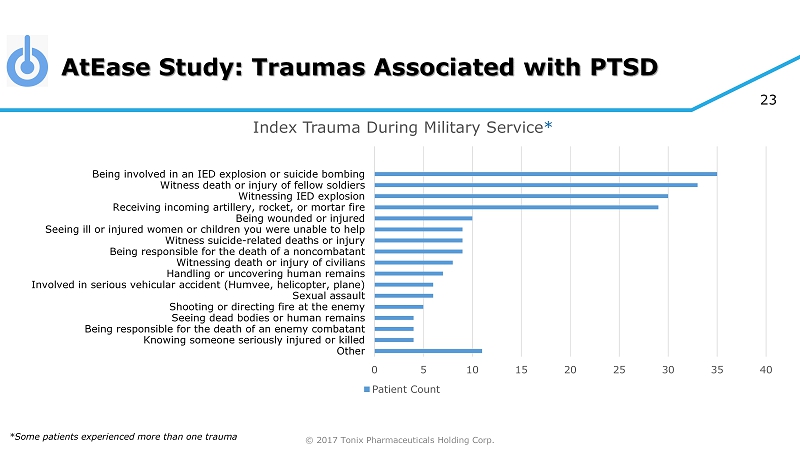
© 2017 Tonix Pharmaceuticals Holding Corp. 23 AtEase Study: Traumas Associated with PTSD *Some patients experienced more than one trauma 0 5 10 15 20 25 30 35 40 Other Knowing someone seriously injured or killed Being responsible for the death of an enemy combatant Seeing dead bodies or human remains Shooting or directing fire at the enemy Sexual assault Involved in serious vehicular accident (Humvee, helicopter, plane) Handling or uncovering human remains Witnessing death or injury of civilians Being responsible for the death of a noncombatant Witness suicide-related deaths or injury Seeing ill or injured women or children you were unable to help Being wounded or injured Receiving incoming artillery, rocket, or mortar fire Witnessing IED explosion Witness death or injury of fellow soldiers Being involved in an IED explosion or suicide bombing Index Trauma During Military Service * Patient Count
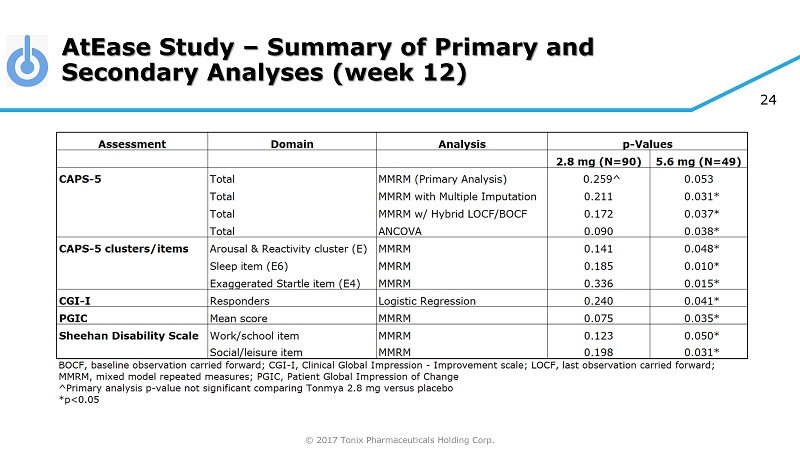
© 2017 Tonix Pharmaceuticals Holding Corp. 24 AtEase Study – Summary of Primary and Secondary Analyses (week 12)
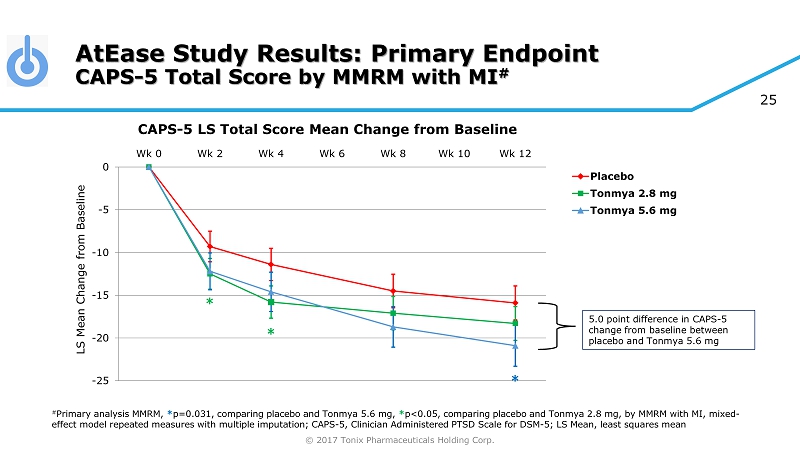
© 2017 Tonix Pharmaceuticals Holding Corp. 25 AtEase Study Results: Primary Endpoint CAPS - 5 Total Score by MMRM with MI # -25 -20 -15 -10 -5 0 Wk 0 Wk 2 Wk 4 Wk 6 Wk 8 Wk 10 Wk 12 Placebo Tonmya 2.8 mg Tonmya 5.6 mg * # Primary analysis MMRM, * p=0.031 , comparing placebo and Tonmya 5.6 mg, * p<0.05, comparing placebo and Tonmya 2.8 mg, by MMRM with MI, mixed - effect model repeated measures with multiple imputation; CAPS - 5, Clinician Administered PTSD Scale for DSM - 5; LS Mean, least squ ares mean * * CAPS - 5 LS Total Score Mean Change from Baseline 5.0 point difference in CAPS - 5 change from baseline between placebo and Tonmya 5.6 mg LS Mean Change from Baseline
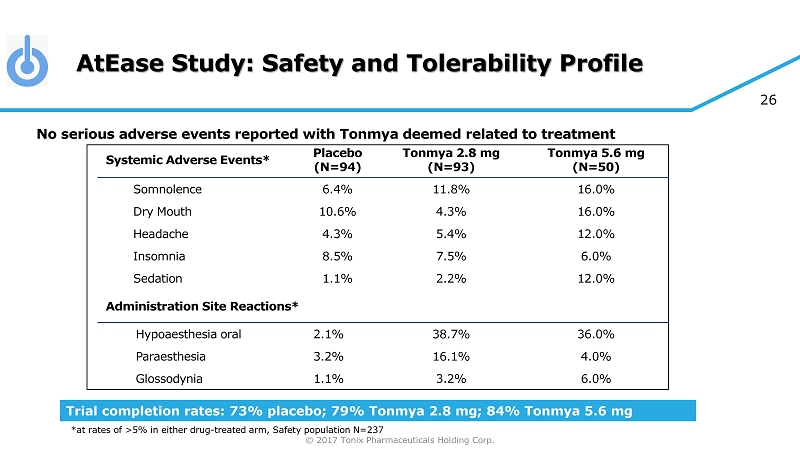
© 2017 Tonix Pharmaceuticals Holding Corp. 26 AtEase Study: Safety and Tolerability Profile No serious adverse events reported with Tonmya deemed related to treatment Systemic Adverse Events* Placebo (N=94) Tonmya 2.8 mg (N=93) Tonmya 5.6 mg (N=50) Somnolence 6.4% 11.8% 16.0% Dry Mouth 10.6% 4.3% 16.0% Headache 4.3% 5.4% 12.0% Insomnia 8.5% 7.5% 6.0% Sedation 1.1% 2.2% 12.0% Administration Site Reactions* Hypoaesthesia oral 2.1% 38.7% 36.0% Paraesthesia 3.2% 16.1% 4.0% Glossodynia 1.1% 3.2% 6.0% Trial completion rates: 73% placebo; 79% Tonmya 2.8 mg; 84% Tonmya 5.6 mg *at rates of >5% in either drug - treated arm, Safety population N=237

© 2017 Tonix Pharmaceuticals Holding Corp. 27 Assessing CAPS - 5 Entry Threshold in AtEase • Score of ≥29 on CAPS - 5 (20 items) required at screening and baseline • >50 on prior versions of CAPS (17 items) typical in previous drug registration trials • Extrapolation from prior versions of CAPS: ((50/17 items)/2) x 20 items = 29.4 • Post - hoc analysis to impute CAPS for DSM - IV ( iCAPS - IV) scores for each subject • Baseline iCAPS - IV score calculated by summing 17 items in common with CAPS - 5 and multiplying by two (for 0 - 8 intensity + frequency rather than 0 - 4) • 4.3% of the sample had baseline iCAPS - IV of ≤ 50 • Choosing CAPS - 5 ≥33 results in all iCAPS - IV > 50 • 80% of mITT had baseline CAPS - 5 of ≥ 33 • Primary analysis of AtEase was run for subgroup with baseline CAPS - 5 ≥ 33
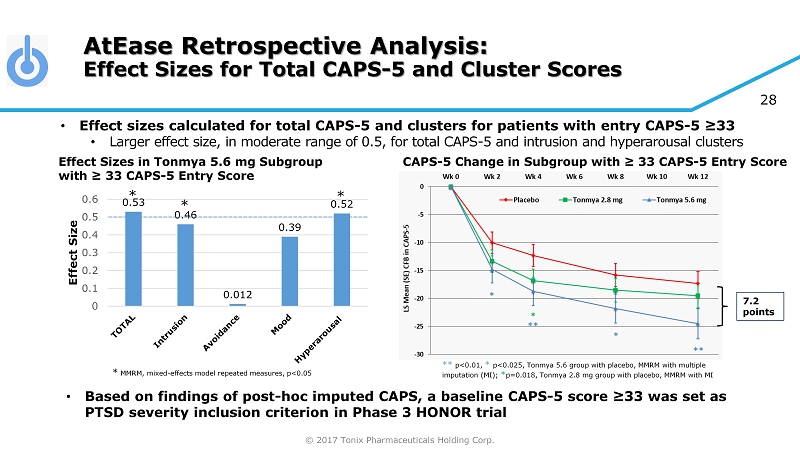
© 2017 Tonix Pharmaceuticals Holding Corp. 28 0.53 0.46 0.012 0.39 0.52 0 0.1 0.2 0.3 0.4 0.5 0.6 Effect Size AtEase Retrospective Analysis: Effect Sizes for Total CAPS - 5 and Cluster Scores * MMRM, mixed - effects model repeated measures, p<0.05 • Effect sizes calculated for total CAPS - 5 and clusters for patients with entry CAPS - 5 ≥33 • Larger effect size, in moderate range of 0.5, for total CAPS - 5 and intrusion and hyperarousal clusters * * * • Based on findings of post - hoc imputed CAPS, a baseline CAPS - 5 score ≥33 was set as PTSD severity inclusion criterion in Phase 3 HONOR trial 7.2 points ** p<0.01, * p<0.025, Tonmya 5.6 group with placebo, MMRM with multiple imputation (MI); * p=0.018, Tonmya 2.8 mg group with placebo, MMRM with MI Effect Sizes in Tonmya 5.6 mg Subgroup with ≥ 33 CAPS - 5 Entry Score CAPS - 5 Change in Subgroup with ≥ 33 CAPS - 5 Entry Score
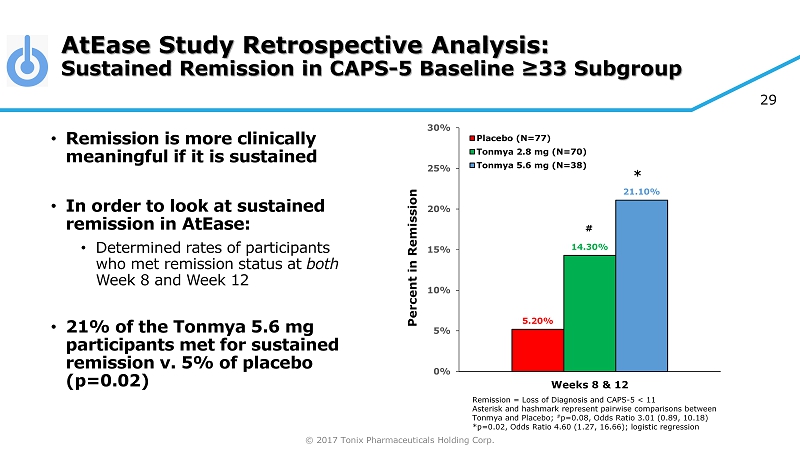
© 2017 Tonix Pharmaceuticals Holding Corp. 29 AtEase Study Retrospective Analysis: Sustained Remission in CAPS - 5 Baseline ≥33 Subgroup Remission = Loss of Diagnosis and CAPS - 5 < 11 Asterisk and hashmark represent pairwise comparisons between Tonmya and Placebo; # p=0.08, Odds Ratio 3.01 (0.89, 10.18) *p=0.02, Odds Ratio 4.60 (1.27, 16.66); logistic regression • Remission is more clinically meaningful if it is sustained • In order to look at sustained remission in AtEase : • Determined rates of participants who met remission status at both Week 8 and Week 12 • 21% of the Tonmya 5.6 mg participants met for sustained remission v. 5% of placebo (p=0.02) 5.20% 14.30% 21.10% 0% 5% 10% 15% 20% 25% 30% Weeks 8 & 12 Percent in Remission Placebo (N=77) Tonmya 2.8 mg (N=70) Tonmya 5.6 mg (N=38) # *
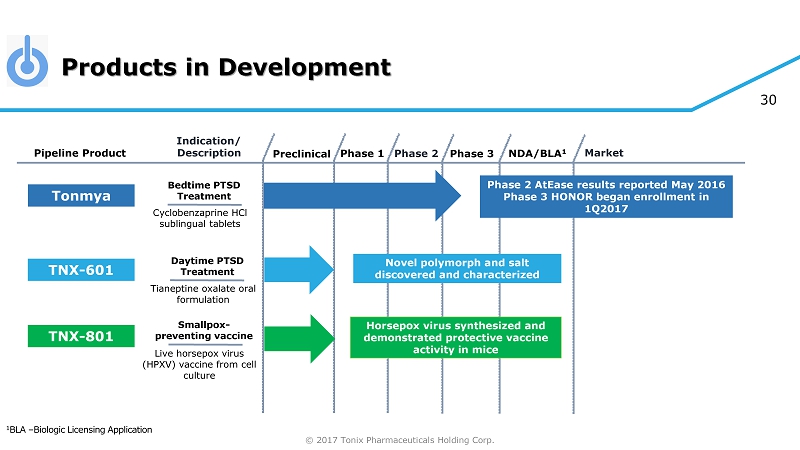
© 2017 Tonix Pharmaceuticals Holding Corp. 30 Products in Development Preclinical Phase 2 NDA/BLA 1 Market Pipeline Product Indication/ Description Phase 3 Tonmya Bedtime PTSD Treatment Daytime PTSD Treatment TNX - 601 Novel polymorph and salt discovered and characterized TNX - 801 Horsepox virus synthesized and demonstrated protective vaccine activity in mice Smallpox - preventing vaccine Cyclobenzaprine HCl sublingual tablets Tianeptine oxalate oral formulation Live horsepox virus (HPXV) vaccine from cell culture Phase 1 Phase 2 AtEase results reported May 2016 Phase 3 HONOR began enrollment in 1Q2017 1 BLA – Biologic Licensing Application

© 2017 Tonix Pharmaceuticals Holding Corp. 31 TNX - 601 ( Tianeptine Oxalate): A Potential Clinical Candidate for PTSD Pre - IND Candidate • Targeted as a 1 st line monotherapy for PTSD : oral formulation for daytime dosing x Leverages expertise in PTSD (clinical and regulatory experience, market analysis, etc.) x Mechanism of Action (MOA) is different from Tonmya • Tianeptine sodium (amorphous) has been approved in EU, Russia, Asia and Latin America for depression since 1987 with established post - marketing experience • Identified new oxalate salt polymorph with improved pharmaceutical properties ideal for reformulation • Filed patent application on novel salt polymorph • Issued patent on steroid - induced cognitive impairment and memory loss issues Targeting a Public Health Challenge • Clinical evidence for PTSD • Several studies have shown tianeptine to be active in the treatment of PTSD 1 - 4 1 Frančišković T, et al. Psychiatr Danub . 2011 Sep;23(3):257 - 63. PMID: 21963693 2 Rumyantseva GM and, Stepanov AL. Neurosci Behav Physiol. 2008 Jan;38(1):55 - 61. PMID: 18097761 3 Aleksandrovskiĭ IA, et al. Zh Nevrol Psikhiatr Im S S Korsakova . 2005;105(11):24 - 9. PMID: 16329631 [Russian] 4 Onder E, et al. Eur Psychiatry. 2006 (3):174 - 9. PMID: 15964747
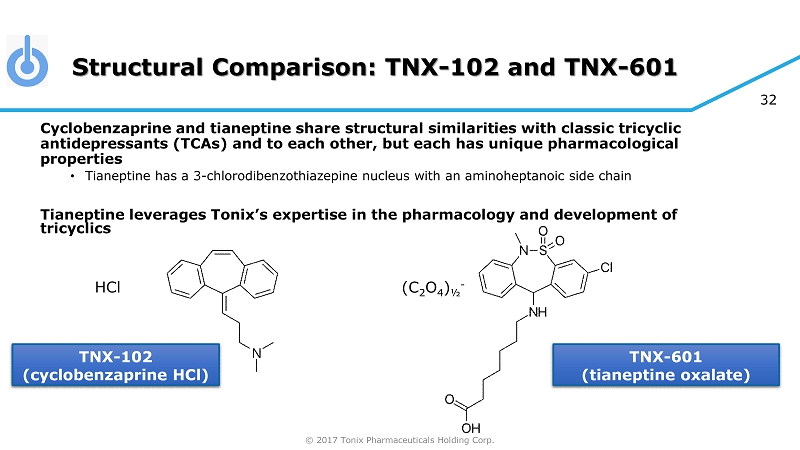
© 2017 Tonix Pharmaceuticals Holding Corp. 32 Structural Comparison: TNX - 102 and TNX - 601 Cyclobenzaprine and tianeptine share structural similarities with classic tricyclic antidepressants (TCAs) and to each other, but each has unique pharmacological properties • Tianeptine has a 3 - chlorodibenzothiazepine nucleus with an aminoheptanoic side chain Tianeptine leverages Tonix’s expertise in the pharmacology and development of tricyclics TNX - 102 (cyclobenzaprine HCl) TNX - 601 (tianeptine oxalate) HCl (C 2 O 4 ) ½ -

© 2017 Tonix Pharmaceuticals Holding Corp. 33 • Tianeptine modulates the glutamatergic system indirectly • Does not have significant affinity (K i >10 μ M) for NMDA or AMPA receptors • Tianeptine is a weak µ - opioid receptor (MOR) agonist • Controlled substance in France, Bahrain and Singapore • Tianeptine has been shown to oppose deleterious effects of chronic stress on brain structure and plasticity TNX - 601: A Potential Clinical Candidate for PTSD • TNX - 601: Novel oxalate salt and polymorph of tianeptine • Improved stability, consistency and manufacturability • Benefited from human experience established in ex - U.S. approved countries • Potential safety and efficacy evidence in published PTSD studies 2 - 5 • 5 year Hatch - Waxman exclusivity for first time approval in the U.S. • Patent filed on novel oxalate salt and polymorph Mechanism of Action Important Characteristics of TNX - 601 1 McEwen, Bruce S., et al. The neurobiological properties of tianeptine (Stablon): from monoamine hypothesis to glutamatergic m odu lation. Molecular psychiatry 2010; 15.3: 237 - 249 2 Frančišković T, et al. Psychiatr Danub . 2011 Sep;23(3):257 - 63. PMID: 21963693 3 Rumyantseva GM and, Stepanov AL. Neurosci Behav Physiol. 2008 Jan;38(1):55 - 61. PMID: 18097761 4 Aleksandrovskiĭ IA, et al. Zh Nevrol Psikhiatr Im S S Korsakova . 2005;105(11):24 - 9. PMID: 16329631 [Russian] 5 Onder E, et al. Eur Psychiatry. 2006 (3):174 - 9. PMID: 15964747 NMDA; N - methyl - D - aspartate, AMPA; α - amino - 3 - hydroxy - 5 - methyl - 4 - isoxazolepropionic acid The active pharmaceutical ingredient (API) is a novel oxalate salt of tianeptine • Shares structural features with tricyclic antidepressants, but has unique pharmacological and neurochemical properties 1 • GMP synthesis developed

© 2017 Tonix Pharmaceuticals Holding Corp. 34 TNX - 801 ( Synthesized Live Horsepox Virus ): A Potential Smallpox - Preventing Vaccine Pre - IND Candidate Potential improvement over current methods for biodefense against smallpox x Leverages government affairs effort x Collaboration with Professor David Evans and Dr. Ryan Noyce at University of Alberta x Protective vaccine activity in mice has been demonstrated x Patent application on novel vaccine submitted Regulatory strategy • FDA’s “Animal Rule” can be applied to establish human efficacy • Good Manufacturing Practice (GMP) virus in development for human safety study Targeting a Public Health Issue Material threat medical countermeasure under 21 st Century Cures Act • Qualifies for receiving Priority Review Voucher on approv al x Priority Review Vouchers have no expiration date, are transferrable and have sold for ~$125 M • ACAM2000 vaccine developed by Acambis was acquired by Sanofi in 2008 for $513 M x ACAM2000 was sold to U.S. Strategic National Stockpile 1 1 Nalca , A et al. Drug design, development and Therapy: 4:71 - 79 (2010)

© 2017 Tonix Pharmaceuticals Holding Corp. 35 TNX - 801 ( Synthesized Live Horsepox Virus ): A Potential Smallpox - Preventing Vaccine Synthesis from sequence of a 1976 Mongolian isolate 1 In mice, TNX - 801 behaved like attenuated vaccinia virus (vaccinia virus is foundation of current smallpox vaccines) How is horsepox related to modern vaccines? • Multiple sources 2 - 4 indicate that the smallpox vaccine discovered by Dr. Edward Jenner in the early 19 th century was likely a horsepox virus • Evidence indicates that modern smallpox vaccines descend from a HPXV - like ancestral strain • Horsepox is now believed to be extinct 4 1 Tulman et al., Journal of Virology, 2006; 80(18): 9244 - 9258 2 Qin et al., Journal of Virology, 2011; 85(24):13049 - 13060 3 Medaglia et al., Journal of Virology, 2015; 89(23):11909 - 11925 4 Esparza J. Veterinary Record. 2013; 173: 272 - 273

© 2017 Tonix Pharmaceuticals Holding Corp. 36 Horsepox - Better Tolerability as a Vaccine? Horsepox is caused by HPXV and is characterized by mouth and skin eruptions HXPV isolate from the 1976 outbreak later sequenced Modern vaccinia virus smallpox vaccines are associated with cardiotoxicity; may have acquired undesirable properties not present in an ancestral virus (likely HPXV) Horsepox has potential for slower proliferation or decreased toxicity

© 2017 Tonix Pharmaceuticals Holding Corp. 37 A Better Smallpox - Preventing Vaccine is Important and Necessary Today Smallpox was eradicated as a result of global public health campaigns No cases of naturally - occurring smallpox have been reported since 1977 Accidental or intentional transmission of smallpox does not require a natural reservoir Stockpiles of smallpox - preventing vaccines are currently maintained and refreshed in case of need

© 2017 Tonix Pharmaceuticals Holding Corp. 38 TNX - 801: A Potential Medical Countermeasure 21st Century Cures Act (2016), Section 3086 • Encouraging treatments for agents that present a national security threat Medical countermeasures are drugs or vaccines intended to treat: • Biological, chemical, radiological, or nuclear agents that present a national security threat • Harm from a condition that may be caused by administering a drug or biological product against such an agent New Priority Review Voucher program for “material threat medical countermeasures” • Priority Review Voucher may be transferred or sold

© 2017 Tonix Pharmaceuticals Holding Corp. 39 Live virus vaccines stimulate cross - reactive immunity • Protects from consequences of infection with smallpox agent • Renders recipient “immune” • Provides additional protection of non - immunized population Potential safety advantage over existing vaccines • Cardiotoxicity limits use of existing vaccines Exclusivity • Patent filed on novel virus composition • Anticipate 12 years exclusivity Mechanism of Action Important Characteristics of TNX - 801 Synthesized live virus HPXV TNX - 801 • Shares structural characteristics with vaccinia - based vaccines • Unique properties that suggest lower toxicity • Smallpox has a ~30% mortality rate in vaccine - naïve populations TNX - 801 ( Synthesized Live Horsepox Virus ): A Potential Smallpox - Preventing Vaccine

© 2017 Tonix Pharmaceuticals Holding Corp. 40 Management Team Seth Lederman, MD President & CEO Jessica Morris EVP, Operations Gregory Sullivan, MD Chief Medical Officer Bradley Saenger, CPA Chief Financial Officer

© 2017 Tonix Pharmaceuticals Holding Corp. 41 Board of Directors Seth Lederman, MD Chairman Ernest Mario, PhD ALZA, Glaxo, Reliant Pharma John Rhodes NYSERDA, NRDC, Booz Allen Hamilton Samuel Saks, MD Jazz Pharma, ALZA, Johnson & Johnson Charles Mather BTIG, Janney, Jefferies, Cowen, Smith Barney Stuart Davidson Labrador Ventures, Alkermes , Combion Patrick Grace Apollo Philanthropy, WR Grace, Chemed Donald Landry, MD, PhD Chair of Medicine, Columbia University

© 2017 Tonix Pharmaceuticals Holding Corp. 42 Milestones – Recently Completed and Upcoming T onmya – Posttraumatic Stress Disorder □ May 2016 Report ed results from Phase 2 AtEase study □ August 2016 End - of - Phase 2 meeting with FDA - Proposed Phase 3 clinical and NDA plan accepted □ December 201 6 Breakthrough Therapy designation granted by FDA □ January 201 7 FDA concurrence with Phase 3 HONOR study design in military - related PTSD □ 1Q 2017 Initial Cross - disciplinary Breakthrough Meeting with FDA □ 1Q 2017 Commenced enrollment of HONOR study □ 2 Q 2017 U.S. Patent No. 9,636,408 issued for eutectic formulation of Tonmya □ 1 H 201 8 Anticipated interim analysis of HONOR study in ~ 275 randomized participants □ 2 H 201 8 Anticipated topline results of HONOR study in 550 participants (if needed) x x x x x x x

© 2017 Tonix Pharmaceuticals Holding Corp. 43 Summary Strong position for value growth with Phase 3 development in a major medical indication: PTSD including military - related PTSD • PTSD is an important public health issue Tonmya for PTSD is designated as a Breakthrough Therapy by FDA • Accelerated development and approval process is expected Phase 3 HONOR study in military - related PTSD began enrollment in 1Q 2017 • Outcome of the interim analysis on ~275 randomized participants (50% efficacy evaluable) expected to be available 1 H 201 8 • Fully funded through the 100% completion of the 550 - participant trial, if needed, and announcement of topline results expected in 2H 2018

© 2017 Tonix Pharmaceuticals Holding Corp. Thank you ! NASDAQ: TNXP